AP* Chemistry
CHEMICAL EQUILIBRIA:
GENERAL CONCEPTS
*AP is a registered trademark of the College Board, which was not involved in the production of, and does not endorse, this
product.© 2008 by René McCormick. All rights reserved.
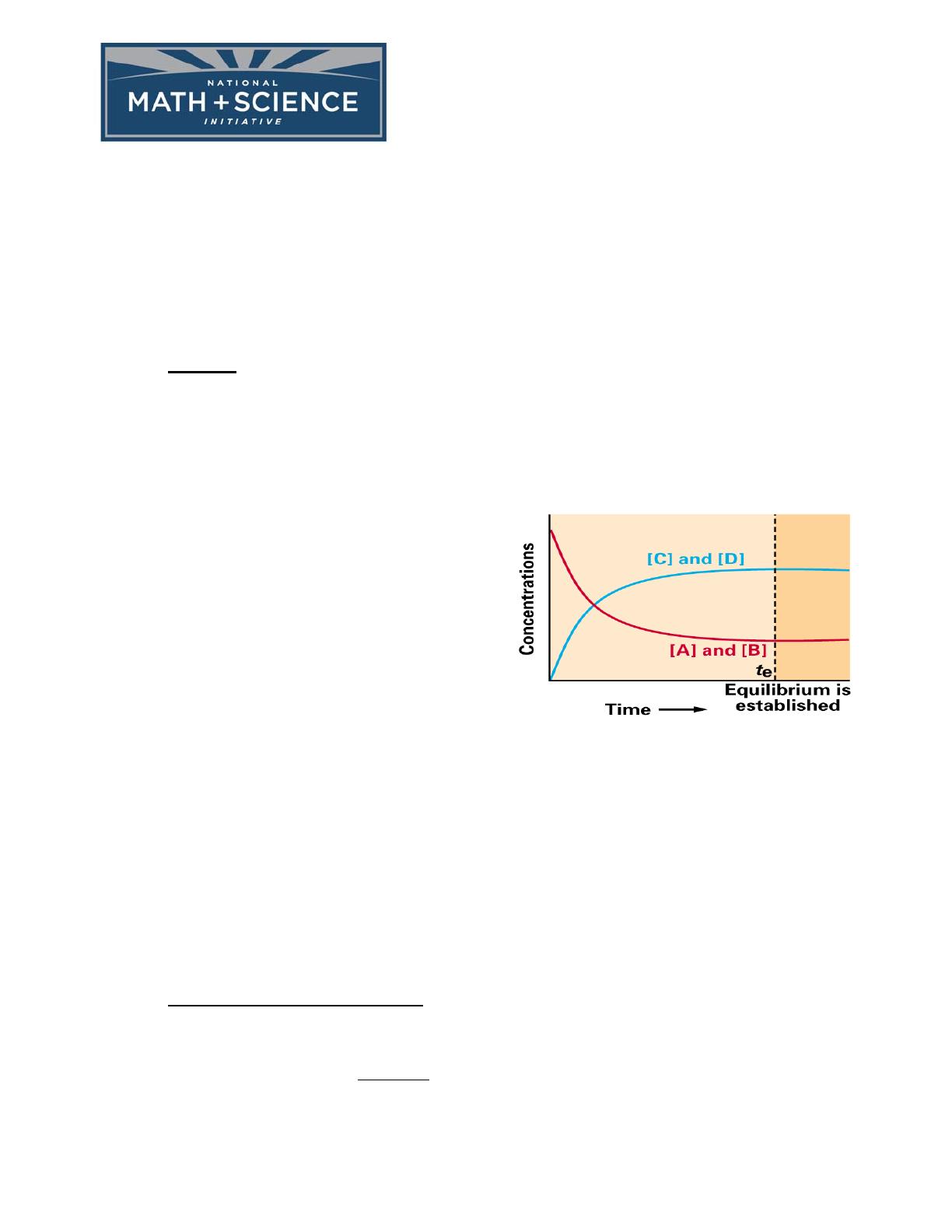
THE NATURE OF THE EQUILIBRIUM STATE: Equilibrium is the state where the rate
of the forward reaction is equal to the rate of the reverse reaction. At these conditions,
concentrations of all reactants and products remain constant with time once equilibrium
has been established at constant temperature. (In stoichiometry, we dealt with equations that
went to completion; often equilibrium equations are going to fall short of this goal.)
Reactions are reversible. This is indicated by double arrows. R
• dynamic--R indicates that the reaction is proceeding in the forward and in the reverse
direction and once equilibrium is established, the rate of each direction is equal. This
keeps the concentration of reactants and products equal.
• the nature and properties of the equilibrium state are the same, no matter what the
direction of approach.
• Examples: Look at the following plot of the reaction between steam and carbon
monoxide in a closed vessel at a high temperature where the reaction takes place rapidly.
H
2
O(g) + CO(g)
R H
2
(g)
+ CO
2
(g)
THE EQUILIBRIUM POSITION: Whether the reaction lies far to the right or to the left
depends on three main factors.
• Initial concentrations (more collisions--faster reaction)
• Relative energies of reactants and products (nature goes to minimum energy)
• Degree of organization of reactants and products (nature goes to maximum disorder)
• The significance of K: K > 1 means that the reaction favors the products at equilibrium
K < 1 means that the reaction favors the reactants at equilibrium
THE EQUILIBRIUM EXPRESSION: A general description of the equilibrium condition
proposed by Gudberg and Waage in 1864 is known as the Law of Mass Action. Equilibrium is
temperature dependent, however, it does not change with concentration or pressure.
• equilibrium constant expression--for the general reaction
aA + bB R cC + dD
Equilibrium constant: K = [C]
c
[D]
d
* Note* K, K
c
, K
eq
may all be used here!
[A]
a
[B]
b




