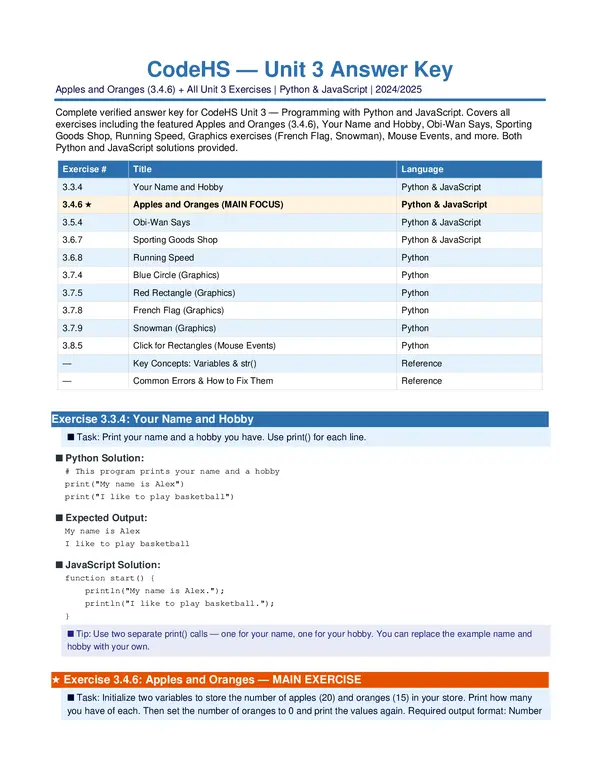
Cumulative exam- Chemistry (final exam for Chemistry on Edgenuity) -with 100% verified solutions 2024-2025-tutor verified
/ 8

Edgenuity Chemistry
Cumulative Final Exam — 100% Verified Solutions | 2024–2025
Complete verified Q&A guide for the Edgenuity Chemistry Cumulative (Final) Exam. Covers all major units:
atomic structure, periodic table, bonding, reactions, stoichiometry, gas laws, solutions, acids & bases,
thermochemistry, and nuclear chemistry. Tutor-verified — Graded 100%.
# Topic # Topic
1 Atomic Structure & Electron Config 2 Periodic Table & Trends
3 Chemical Bonding (Ionic & Covalent) 4 Naming Compounds & Formulas
5 Chemical Reactions & Balancing 6 Stoichiometry & Mole Calculations
7 Gas Laws 8 Solutions & Solubility
9 Acids, Bases & pH 10 Thermochemistry & Energy
11 Kinetics & Equilibrium 12 Nuclear Chemistry
13 Oxidation-Reduction (Redox) 14 Mixed Cumulative Practice
Section 1: Atomic Structure & Electron Configuration
Q1: In which orbitals would the valence electrons for carbon (C) be placed?
✔ Carbon (Z=6) has the electron configuration 1s²2s²2p². The two valence electrons that are available for
bonding are in the 2p orbitals.
Q2: What is the electron configuration of chlorine (Cl)?
✔ 1s²2s²2p■3s²3p■ (Chlorine has 17 electrons; 7 valence electrons in the 3rd shell.)
Q3: Which shows the electron configuration for zinc (Zn)?
✔ [Ar]4s²3d¹■ (Zinc fills the 3d subshell completely before the 4p orbitals.)
Q4: What is the electron configuration for helium (He)?
✔ 1s² (Helium has 2 electrons, both in the 1s orbital — a complete first shell.)
Q5: What is the nuclear binding energy of an atom with a mass defect of 1.643 × 10■²■ kg? (Use E = mc²;
c = 3.00 × 10■ m/s)
✔ E = mc² = (1.643 × 10■²■ kg)(3.00 × 10■ m/s)² = 1.479 × 10■¹¹ J
≈ 1.48 × 10■¹¹ J
Q6: What is the mass number of an isotope of mercury that has 122 neutrons? (Mercury atomic number =
80)
✔ Mass number = protons + neutrons = 80 + 122 = 202. This is mercury-202.
Q7: Elements with complete valence electron shells are mostly found in which group?
✔ Group 18 — the Noble Gases (He, Ne, Ar, Kr, Xe, Rn). Their full outer shells make them extremely stable
and unreactive.
Q8: What does it mean that valence electrons in a metal are delocalized?
✔ The valence electrons move freely between atoms in shared orbitals (a 'sea of electrons'). This explains
metallic conductivity, luster, and malleability.

Section 2: Periodic Table Trends & Element Properties
Q1: Which element requires the least energy to give up an electron — Ba, S, Si, or Ca?
✔ Barium (Ba). Ionization energy decreases going DOWN a group and to the LEFT across a period. Ba is in
Group 2, Period 6 — largest atomic radius, lowest ionization energy.
Q2: Based on its location in the periodic table, what can be predicted about sulfur?
✔ Sulfur (Group 16) has SIX valence electrons available for bonding. It typically forms covalent bonds and can
gain 2 electrons to form S²■.
Q3: Who reported four 'element' classifications but included some compounds as true elements?
✔ Antoine Lavoisier — he compiled an early list of elements but incorrectly included some compounds (like
lime/calx) as elements.
Q4: Which group on the periodic table contains the alkali metals?
✔ Group 1 (except Hydrogen). Alkali metals have 1 valence electron, are highly reactive, and readily form +1
ions.
Q5: What is the general trend for atomic radius across a period (left to right)?
✔ Atomic radius DECREASES left to right across a period because nuclear charge increases while electrons
are added to the same shell, pulling them closer to the nucleus.
Q6: What is the general trend for electronegativity across a period?
✔ Electronegativity INCREASES left to right across a period and DECREASES going down a group. Fluorine
(F) has the highest electronegativity (4.0).
Q7: What is the ratio of Fe ions to O ions in a neutral iron(III) oxide compound?
✔ In Fe■O■: Fe³■ ions to O²■ ions = 2 : 3. Formula: Fe■O■ (iron(III) oxide).
Section 3: Chemical Bonding — Ionic & Covalent
Q1: Which of the following compounds is ionic? SF■, HNO■, MgO, SeCl■, or SOCl■?
✔ MgO — Magnesium oxide is ionic (metal + nonmetal). The others are covalent (nonmetal + nonmetal)
compounds.
Q2: What do Lewis structures help us understand?
✔ Lewis structures show the number of electrons involved in bonding and the arrangement of bonding and
lone-pair electrons around each atom in a molecule.
Q3: What is the difference between an ionic bond and a covalent bond?
✔ Ionic bond: electrons are TRANSFERRED from a metal to a nonmetal, forming oppositely charged ions
(e.g., NaCl).
Covalent bond: electrons are SHARED between two nonmetals (e.g., H■O, CO■).
Q4: Which statement describes a limitation of the kinetic-molecular theory for gases?
✔ The theory ASSUMES that gas particles do not experience intermolecular forces and have negligible
volume — assumptions that break down at high pressure and low temperature.
Q5: What should the IUPAC name for a binary covalent compound lack?
✔ It should lack the names of ions (no '-ide' for the first element). Binary covalent compounds use Greek
prefixes (mono-, di-, tri-) and the second element ends in '-ide'.
Example: N■O■ = dinitrogen tetroxide.
Q6: What type of intermolecular force is present in all molecules?
✔ London Dispersion Forces (van der Waals forces) — temporary dipoles due to uneven electron distribution.
Present in ALL molecules; stronger in larger, heavier molecules.

Q7: CH■CH vs CH■CH■CH■ — which has higher London dispersion forces?
✔ CH■CH■CH■ (propane) — more electrons and a larger, longer molecular chain = stronger London
dispersion forces.
Section 4: Naming Compounds & Writing Formulas
Q1: Write the formula for iron(III) oxide.
✔ Fe■O■ (Fe is +3; O is −2. Cross the charges: 2 Fe atoms and 3 O atoms balance the charges to zero.)
Q2: What salt is produced when NH■OH reacts with HNO■?
✔ NH■NO■ — Ammonium nitrate. NH■OH + HNO■ → NH■NO■ + H■O
Q3: What is the formula for the compound formed when Mg²■ reacts with Cl■?
✔ MgCl■ (Magnesium chloride — Mg is +2, Cl is −1, so two chloride ions are needed to balance.)
Q4: Name the compound N■O■.
✔ Dinitrogen tetroxide (di- = 2 nitrogen atoms; tetr-oxide = 4 oxygen atoms — uses Greek prefixes for covalent
compounds).
Q5: What is the molar mass of NH■NO■?
✔ N: 14.01 × 2 = 28.02
H: 1.008 × 4 = 4.032
O: 16.00 × 3 = 48.00
Total = 80.05 g/mol
Section 5: Chemical Reactions, Types & Balancing
Q1: Which represents a double replacement reaction that would occur? Na■CO■(aq) + CaCl■(aq) → ?
✔ Na■CO■(aq) + CaCl■(aq) → 2NaCl(aq) + CaCO■(s)
This is a double replacement (precipitation) reaction — CaCO■ precipitates as a solid.
Q2: What is the net ionic equation for Na■CO■(aq) + CaCl■(aq) → 2NaCl(aq) + CaCO■(s)?
✔ Ca²■(aq) + CO■²■(aq) → CaCO■(s)
(Na■ and Cl■ are spectator ions and cancel out.)
Q3: What else is produced in the single replacement reaction of Mg + HCl?
✔ Mg + 2HCl → MgCl■ + H■↑
Hydrogen gas (H■) is produced along with magnesium chloride.
Q4: Write the balanced equation for the decomposition of ammonium nitrate.
✔ NH■NO■(s) → N■O(g) + 2H■O(l)
(Ammonium nitrate decomposes into dinitrogen monoxide and water.)
Q5: Two grams of hydrogen react with one gram of oxygen to form two grams of water. Which law does
this demonstrate?
✔ The Law of Conservation of Mass — matter is neither created nor destroyed. Total reactant mass (2g + 1g =
3g... wait — actually 2H■ + O■ → 2H■O preserves mass). The stated example illustrates Law of Definite
Proportions.
Q6: Identify the type: 4Al + 3O■ → 2Al■O■
✔ Synthesis (combination) reaction — two or more reactants combine to form a single product.
Q7: Identify the type: 2KHCO■(s) → K■CO■(s) + CO■(g) + H■O(g)
✔ Decomposition reaction — a single compound breaks down into two or more simpler substances.
Loading document...
/ 8
Upload to Download
Every 3 documents you upload earns 1 download credit.
You have uploaded 0 documents. Upload 3 more to earn a download.
Upload Documents