The document outlines a WAEC Chemistry practical for 2025/2026, focusing on quantitative analysis through titration to determine the concentration of Na2CO3·10H2O using HCl. It includes a balanced chemical equation, titration data, and qualitative analysis tests with observations and inferences for various reagents. Key results indicate the presence of Zn^2+, Cl-, CO3^2-, and reducing sugars in the tested solutions.
/ 2
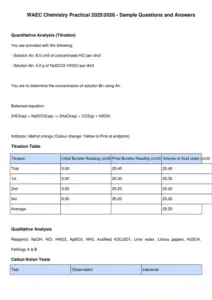
WAEC Chemistry Practical 2025/2026 - Sample Questions and Answers
Quantitative Analysis (Titration)
You are provided with the following:
- Solution An: 8.5 cm3 of concentrated HCl per dm3
- Solution Bn: 5.0 g of Na2CO3·10H2O per dm3
You are to determine the concentration of solution Bn using An.
Balanced equation:
2HCl(aq) + Na2CO3(aq) -> 2NaCl(aq) + CO2(g) + H2O(l)
Indicator: Methyl orange (Colour change: Yellow to Pink at endpoint)
Titration Table
Titration Initial Burette Reading (cm3) Final Burette Reading (cm3) Volume of Acid Used (cm3)
Trial 0.00 25.40 25.40
1st 0.00 25.30 25.30
2nd 0.00 25.20 25.20
3rd 0.00 25.20 25.20
Average 25.20
Qualitative Analysis
Reagents: NaOH, HCl, HNO3, AgNO3, NH3, Acidified K2Cr2O7, Lime water, Litmus papers, H2SO4,
Fehlings A & B
Cation/Anion Tests
Test Observation Inference

Add NaOH to unknown solution White ppt soluble in excess Zn^2+ present
Add NH3 to unknown White ppt soluble in excess Zn^2+ confirmed
Add AgNO3 + HNO3 White ppt formed Cl- present
Add HCl to solid, test gas with lime waterEffervescence, gas turns lime water milkyCO3^2- present
Add acidified K2Cr2O7 Solution turns green Reducing agent present
Add litmus paper Red litmus turns blue Alkaline solution
Fehling's A & B + heat with glucoseBrick-red ppt Reducing sugar present
Loading document...
/ 2
Upload to Download
Every 3 documents you upload earns 1 download credit.
You have uploaded 0 documents. Upload 3 more to earn a download.
Upload Documents