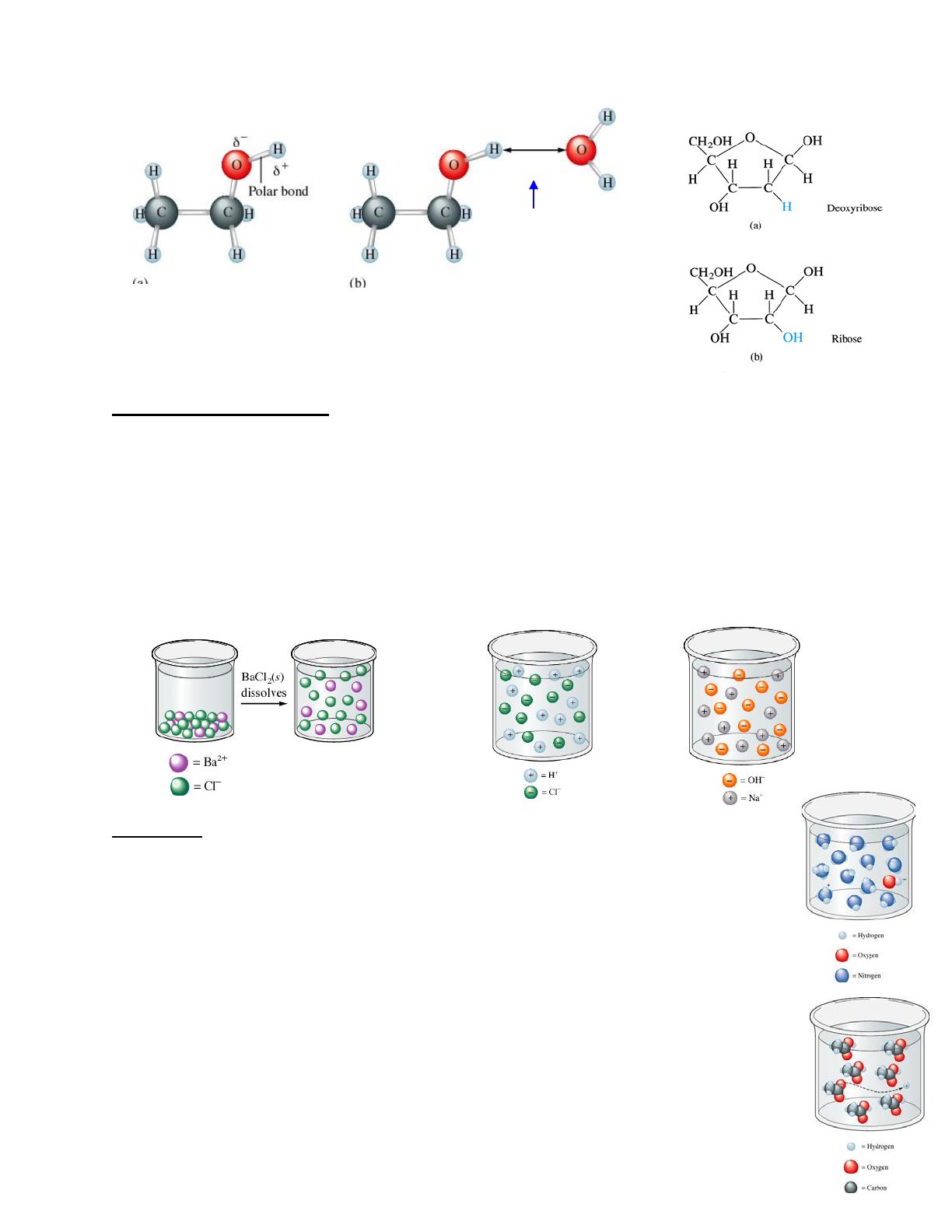
Hydration in action due to
an electrostatic attraction
called a hydrogen bond.
Can also be represented
by a dashed line.
ethanol IS attracted to water
Water can also dissolve nonionic substances such as alcohols and sugars among others. Alcohols and
sugars contain O—H bonds that are polar just as the O—H bond in water. This polarity makes the
molecule soluble.
Fats do not dissolve in water since they are nonpolar.
“Like dissolves like” is a useful guideline for predicting solubility, BUT it is not an explanation!
You must address the electrostatic (“opposites attract”) aspects of solute-solvent interactions.
THE NATURE OF AQUEOUS SOLUTIONS: STRONG AND WEAK ELECTROLYTES
Remember a solution is a homogeneous mixture where a solute is dissolved in a solvent. Aqueous solutions
are solutions where the solvent is water.
Properties of Aqueous Solutions
x electrolytes – solutions that conduct an electric current; the more mobile & charged
particles present, the higher the concentration of ions in solution, the better the
conduction. The magnitude of the charge also matters.
x strong – completely dissociate (consult solubility rules—strong acids, strong bases and
soluble salts)
x Barium chloride is an ionic salt that completely ionizes in water, HCl is a strong acid that
completely dissociates into H
+
ions and Cl
ions in water while NaOH is a strong base that
completely dissociates into Na
+
ions and OH
ions water. All 3 release mobile, charged
particles that readily conduct electricity.
x COMMON Strong acids: HCl, HBr, HI, HNO
3
, H
2
SO
4
[loses first H
+
easily and exists
mainly as H
+
and HSO
4
ions in water], and HClO
4
x COMMON Strong bases: Oxides and Hydroxides of I & II A metals [solubility issues with
the IIA’s]
x weak – do not completely dissociate; only about 1% dissociation (The weak acid, acetic
acid and the weak base ammonia are pictured right. Both are classic examples!)
Types of Reactions & Solution Stoichiometry
2




