AP Chemistry Unit 9 Progress Check focuses on free-response questions that assess students' understanding of thermodynamics, entropy, and redox reactions. This guide provides detailed explanations and calculations related to standard entropy changes, thermodynamic favorability, and oxidation-reduction processes. Designed for AP Chemistry students preparing for the exam, it includes practice problems and scoring criteria. The content aligns with the 2024 AP Chemistry curriculum, ensuring relevance and applicability for exam preparation.
Key Points
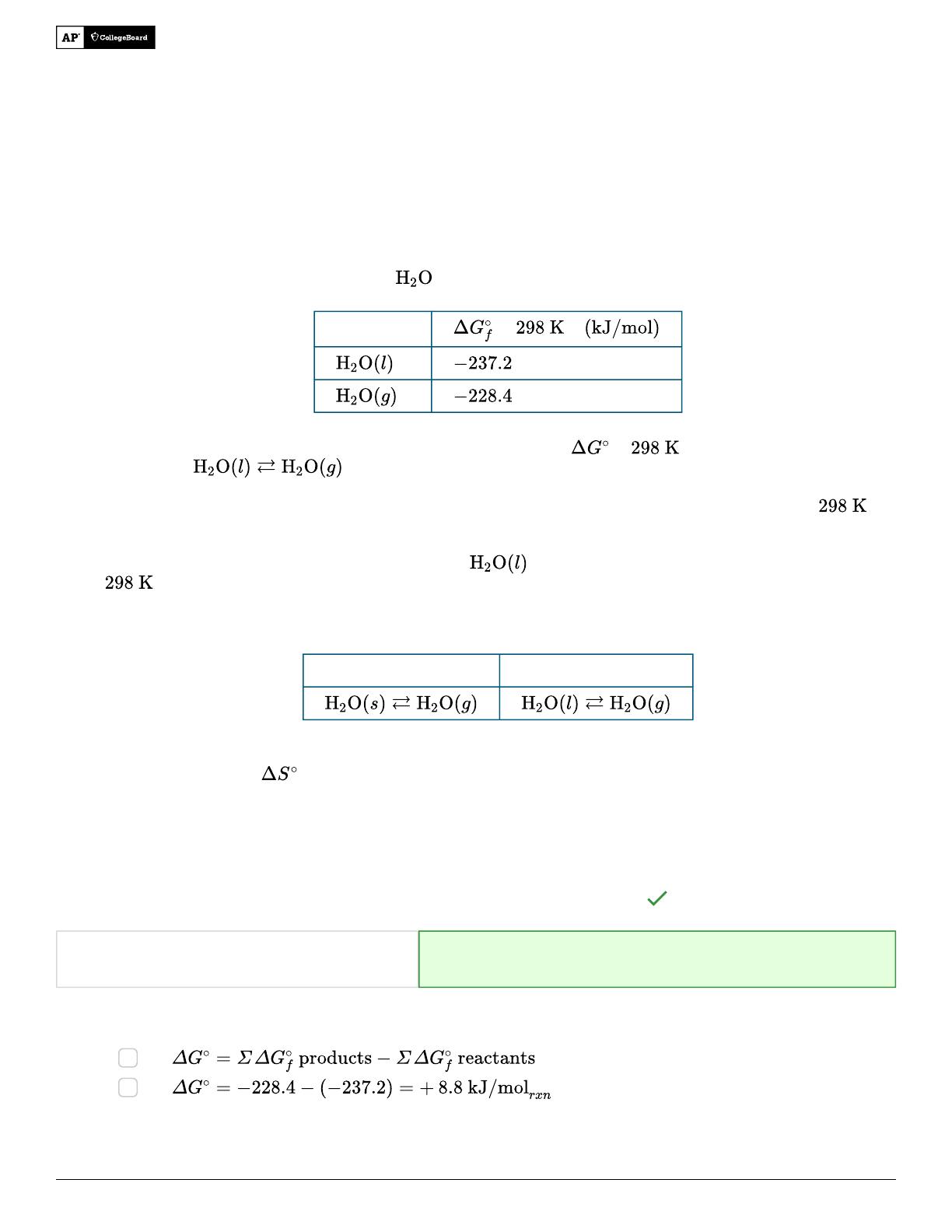
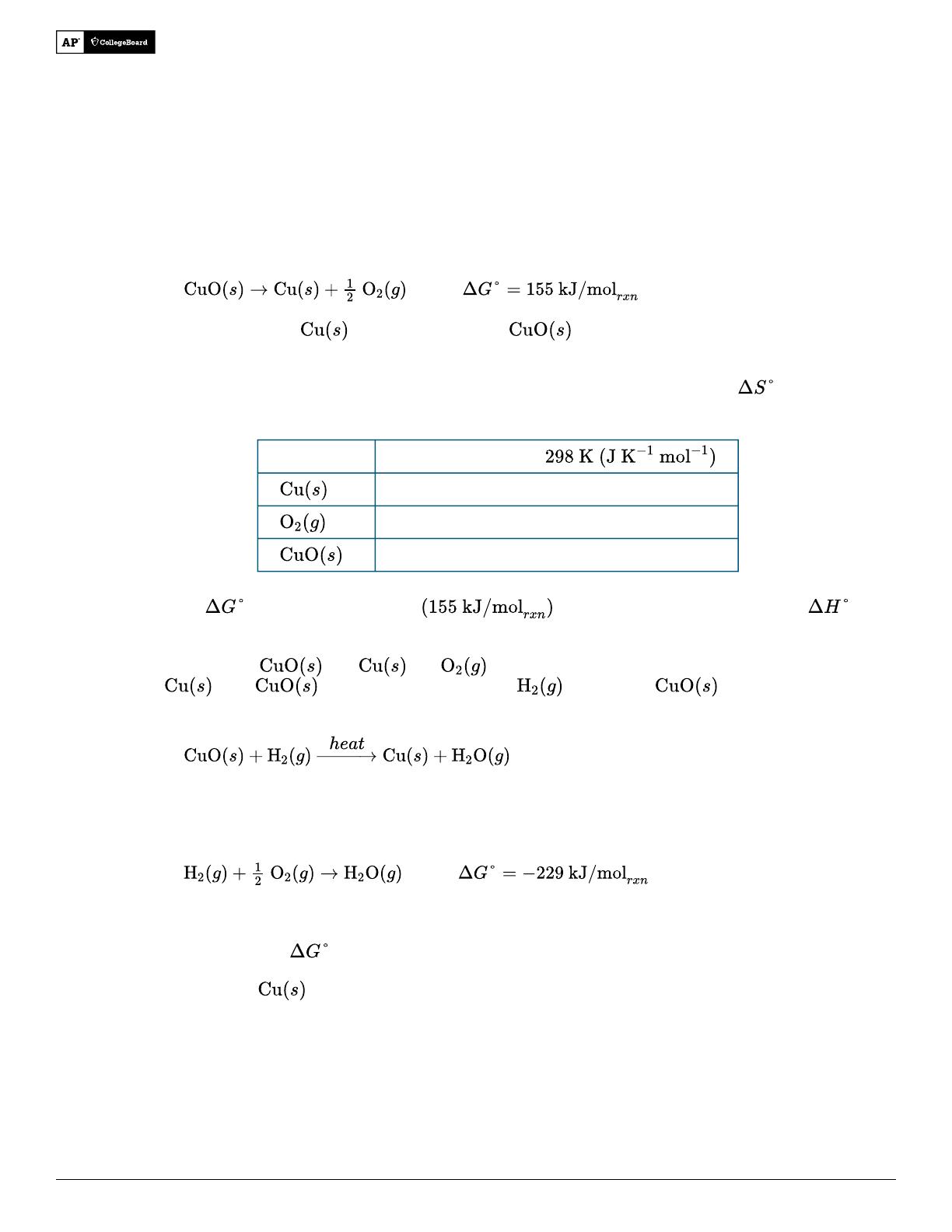
- Analyzes thermodynamic favorability using Gibbs free energy calculations.
- Explains entropy changes in phase transitions of water, including sublimation and vaporization.
- Covers redox reactions with oxidation state changes and half-reaction methods.
- Includes scoring criteria for free-response questions to aid in exam preparation.