CH142 S26 Exam 3 Sample Key provides detailed solutions and explanations for various chemistry problems relevant to the course. This key covers topics such as coordination complexes, solubility, titration, and buffer capacity, which are essential for students preparing for exams in introductory chemistry. It includes calculations related to Ksp values and the behavior of acids and bases, making it a valuable resource for students seeking to enhance their understanding of chemical principles. Ideal for CH142 students looking to review key concepts and practice problem-solving skills.
Key Points
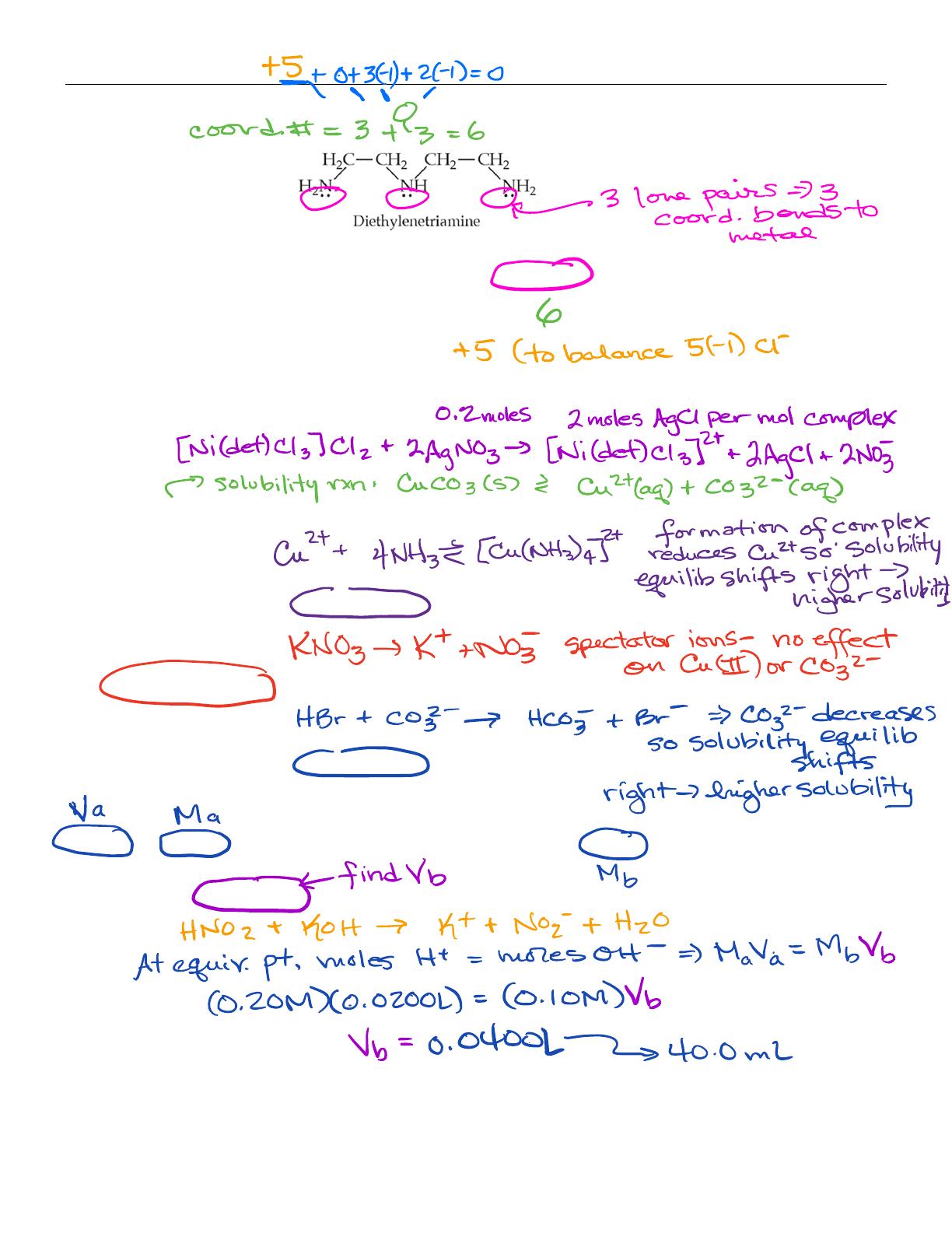
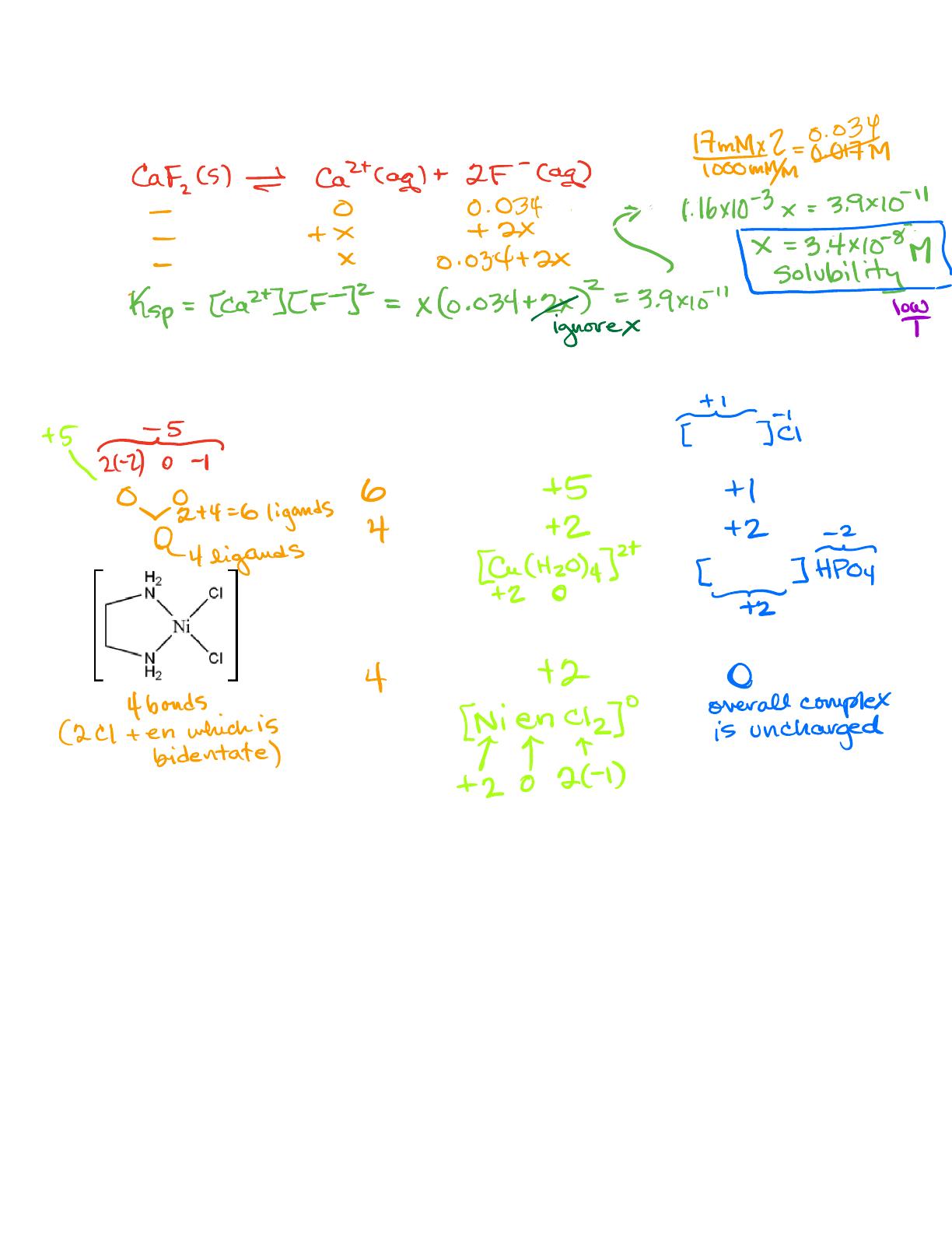
- Includes solutions for coordination complex problems and their properties.
- Covers solubility calculations for compounds like CaF2 in different conditions.
- Explains the titration process for weak acids and bases, including pH changes.
- Discusses buffer capacity and its implications for biochemical applications.