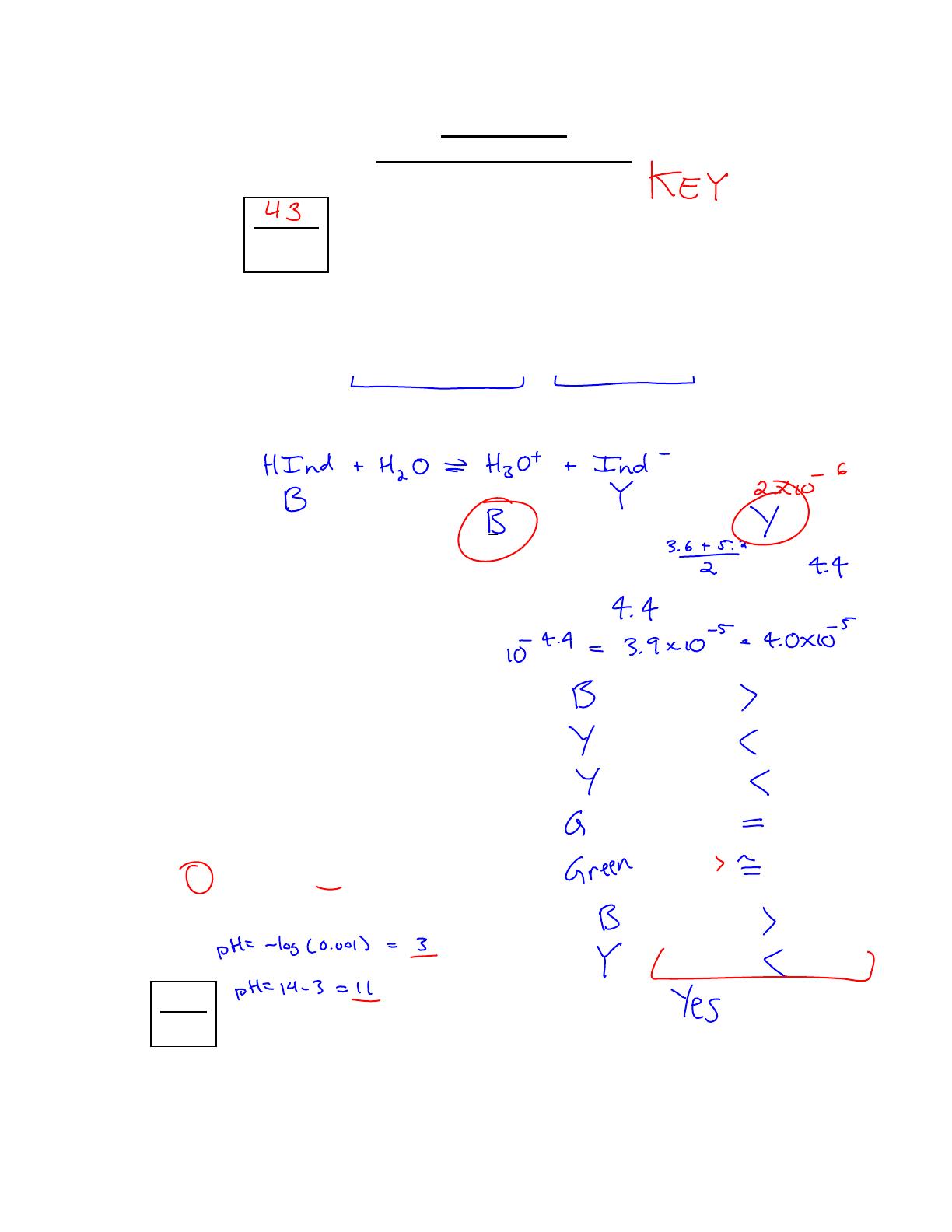
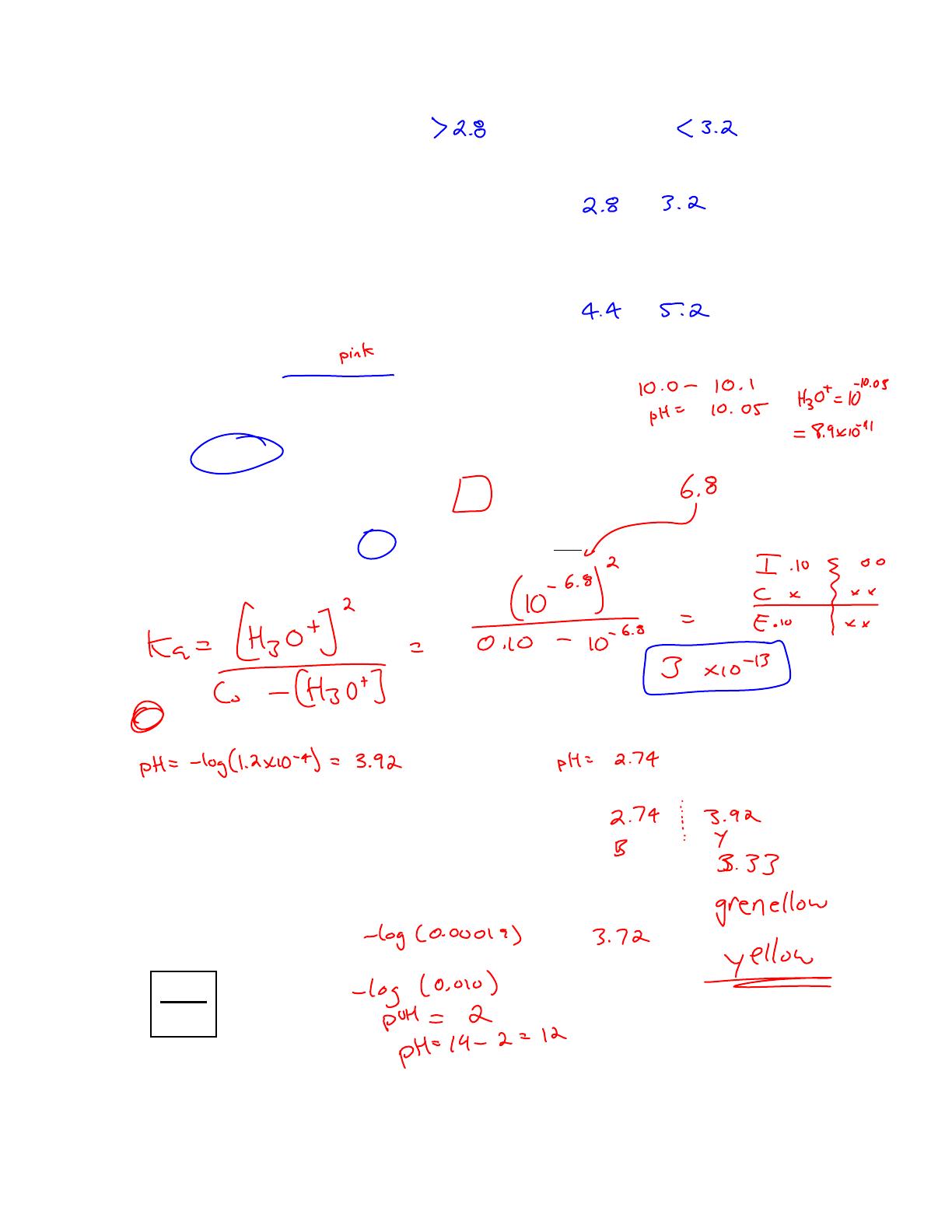
Chemistry 12 Worksheet 4-7 focuses on indicators, detailing the behavior of various indicators in different pH environments. This answer key provides solutions to questions about the color changes of indicators like HInd, Orange IV, and methyl orange, as well as calculations for pKa and Ka values. It is designed for high school chemistry students preparing for exams and understanding acid-base chemistry. The worksheet includes equilibrium equations and analysis of weak acids, making it a valuable resource for mastering indicator concepts.
Key Points
- Includes answers for Chemistry 12 Worksheet 4-7 on indicators.
- Covers pH ranges and color changes for various indicators.
- Provides calculations for pKa and Ka values of weak acids.
- Explains the equilibrium equations for indicators like HInd.