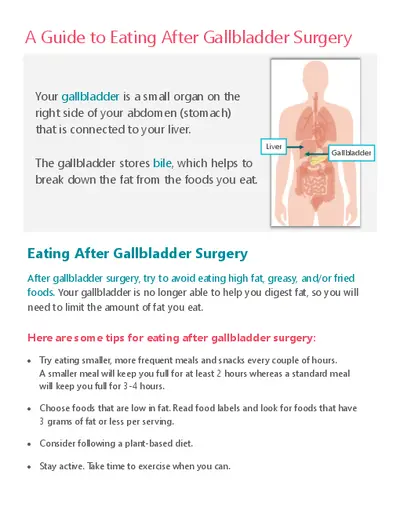
PLASMODIUM SPP.
Malaria remains the leading parasitic disease that causes
mortality worldwide.
This disease is caused by malaria parasites which are
micro-organisms that belong to the genus Plasmodium that
is transmitted by the bite of an infected female mosquito
belonging to the genus Anopheles.
The known species infecting humans:
P. falciparum
P. vivax
P. ovale
P malariae
P. knowlesi
P. vivax – the widest geographic distribution and is the
species most likely to be found in temperate climates.
P. vivax and P. falciparum – cause more than 95% of
infections.
P. ovale – primarily confined to western parts of Africa.
P. knowlesi – causes malaria in the macaque monkey, has
also been implicated in cases of zoonotic human malaria in
Malaysia and the Philippines.
In general, infections caused by P. vivax, P. ovale, and P.
malariae are less severe than those caused by P.
falciparum.
Life Cycle
Ring-Form Trophozoite (early trophozoite) – In human
infections, this is the earliest stage, in which the organism
has a prominent, red to purple chromatin dot and a small
blue ring of cytoplasm surrounding a vacuole.
Growing Trophozoite (Late Trophozoile) – characterized
by an increase in the amount of cytoplasm, the
disappearance of the vacuole, and the appearance of
malarial pigment in the organism's cytoplasm.
Immature Schizont (Multinucleate Stage) –
characterized by a splitting of the chromatin mass.
Mature Schizont – contains merozoites, which are
individual chromatin masses, each surrounded by
cytoplasm.
Microgametocytes (Male) – have pale blue cytoplasm and
a diffuse chromatin mass that stains pale pink to purple.The
chromatin may be surrounded by a clear halo.
Macrogametocytes (Female) – show a well-defined,
compact chromatin mass that stains dark pink; the
cytoplasm stains a darker blue than microgametocytes
Exoerythrocytic Phase
Humans serve as intermediate hosts and acquire the
infection when the female mosquito takes a blood meal and
injects the infective sporozoites with salivary secretions.
The sporozoites enter the human circulation and take
approximately 60 minutes to reach the liver, where they
begin the exoerythrocytic phase by penetrating
parenchymal cells.
Maturation through the trophozoite and schizont phases
results in the production of hepatic merozoites.
The release of mature merozoites from liver cells and
invasion of RBCs signal the beginning of the erythrocytic
phase.
Generally, only one cycle of merozoite production occurs in
ther liver before RBCs are invaded.
P. vivax and P. ovale, however, may persist in the liver in a
dormant stage known as hypnozoites, which accounts for
the relapse (recurrence) of the disease within 1 to 3 years
after the primary infection.
Erythrocytic Phase
Merozoites have structures and proteins (e.g., erythrocyte-
binding antigen) that selectively adhere to receptors on the
RBC membrane.
P. vivax, as well as P. knowlesi, use antigens of the
Duffy blood group as receptors for attachment to
and internalization into the RBCs, whereas P.
falciparum may simply attach to receptors that are
integral parts of the RBC membrane itself.
When merozoites have attached, endocytic invagination of
the RBC membrane allows the organism to enter the RBC
within a vacuole.
Once inside the RBC, the organism feeds on hemoglobin and
initiates the erythrocytic phase.
Malarial pigment (hemozoin), which is composed of iron
deposits, is formed in the growing trophozoite as a result of
incomplete metabolism of hemoglobin.
Once the organism has reached the mature trophozoite
stage, the chromatin begins to divide (developing schizont).