Part B: Chromatography of Amino Acids
Paper chromatography separates amino acids based on their polarity and interaction with the stationary
phase (paper) and mobile phase (solvent). The R
f
value (retention factor) is calculated to identify each
amino acid.
R
f
Value Calculation
R
f
= Distance traveled by amino acid / Distance traveled by solvent front
Practice Problem:
A student calculated that the solvent front was 8.0 cm above the starting line. Arginine traveled 2.0 cm
and glycine traveled 4.0 cm . Calculate the R
f
values.
Answers:
Amino Acid Distance
Traveled
Solvent Front R
f
Calculation R
f
Value
Arginine 2.0 cm 8.0 cm 2.0 / 8.0 0.25
Glycine 4.0 cm 8.0 cm 4.0 / 8.0 0.50
Which amino acid traveled further? Why?
Glycine traveled further (4.0 cm vs. 2.0 cm). Glycine is a smaller, less polar amino acid with a
simple hydrogen side chain (–H), so it interacts less with the polar stationary phase (paper) and
moves more readily with the mobile solvent. Arginine is a larger, basic amino acid with a positively
charged guanidinium group in its side chain at neutral pH. This polar, charged side chain causes
stronger interactions with the polar paper, slowing its movement and resulting in a lower R
f
value.
Part C: Analysis Questions with Answers
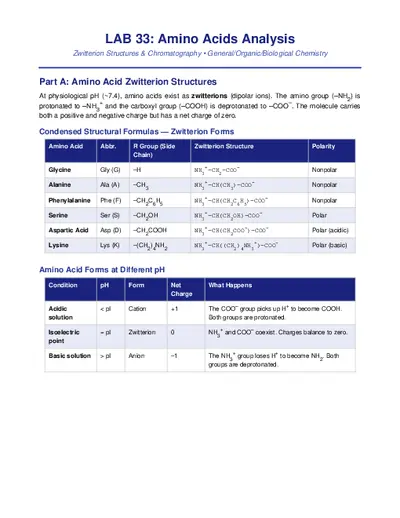
Q1. What is a zwitterion?
A zwitterion is a molecule that contains both a positive charge and a negative charge on different
atoms but has an overall net charge of zero. In amino acids, the amino group is protonated (NH
3
+
)
and the carboxyl group is deprotonated (COO
–
). Zwitterions are also called dipolar ions or internal
salts.
Q2. Why do amino acids have high melting points compared to other organic compounds of
similar size?
Amino acids exist as zwitterions (internal salts) with strong electrostatic attractions between the
positive NH
3
+
and negative COO
–
groups of neighboring molecules. These ionic interactions are
much stronger than the van der Waals forces or hydrogen bonds found in similarly sized non-ionic
organic compounds, requiring more energy (higher temperature) to overcome.
Q3. Draw the structure of glycine in: (a) acidic solution, (b) neutral/zwitterion form, (c) basic
solution.