Chemical equilibrium is a fundamental concept in chemistry that describes the state of a reversible reaction when the rates of the forward and reverse reactions are equal. This lecture covers key principles such as dynamic equilibrium, the equilibrium constant (K), and the factors affecting equilibrium, including concentration, pressure, and temperature changes. Designed for students studying general chemistry, it provides a comprehensive overview of the laws governing chemical reactions. The material includes practical applications and examples to illustrate how equilibrium concepts are used in real-world scenarios.
Key Points
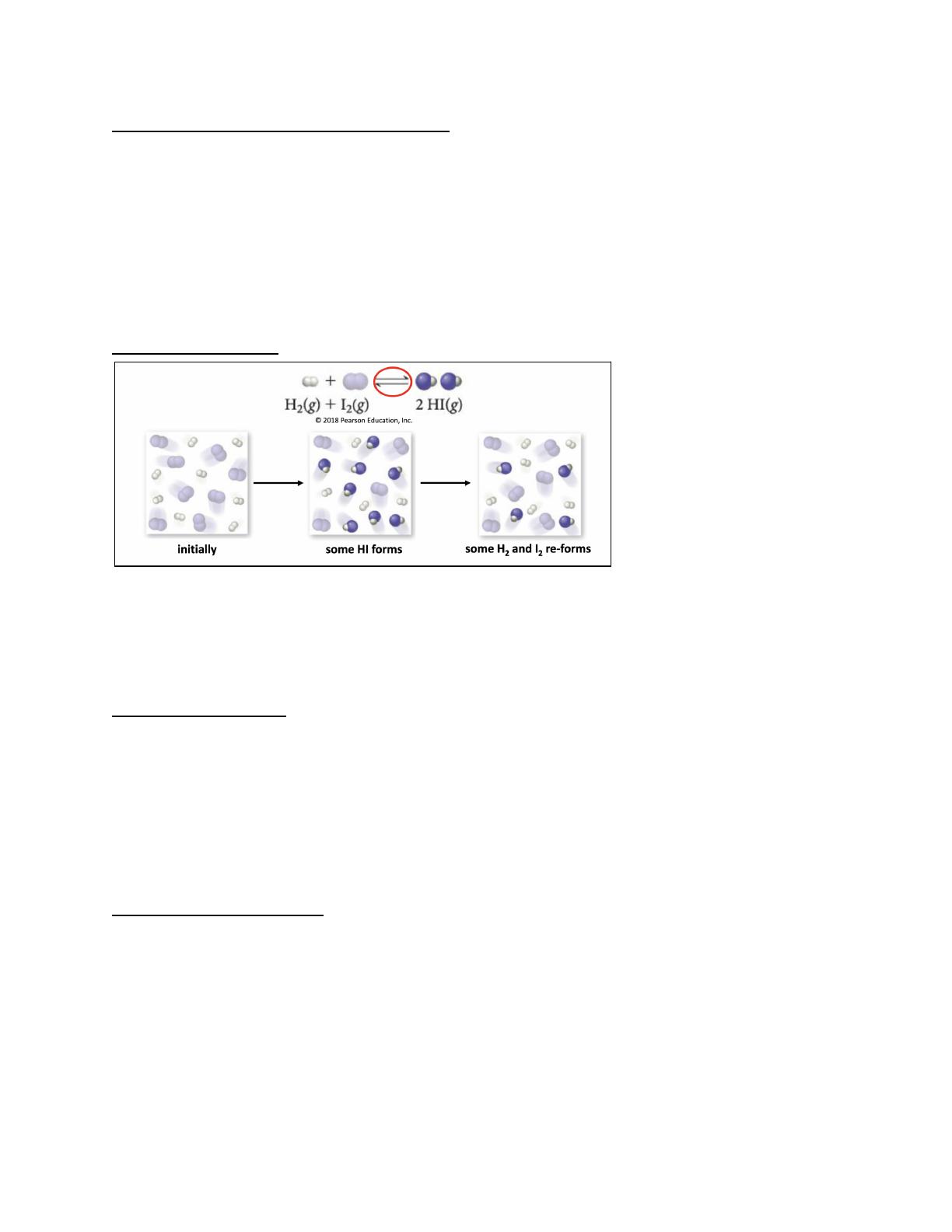
- Explains dynamic equilibrium and its significance in reversible reactions.
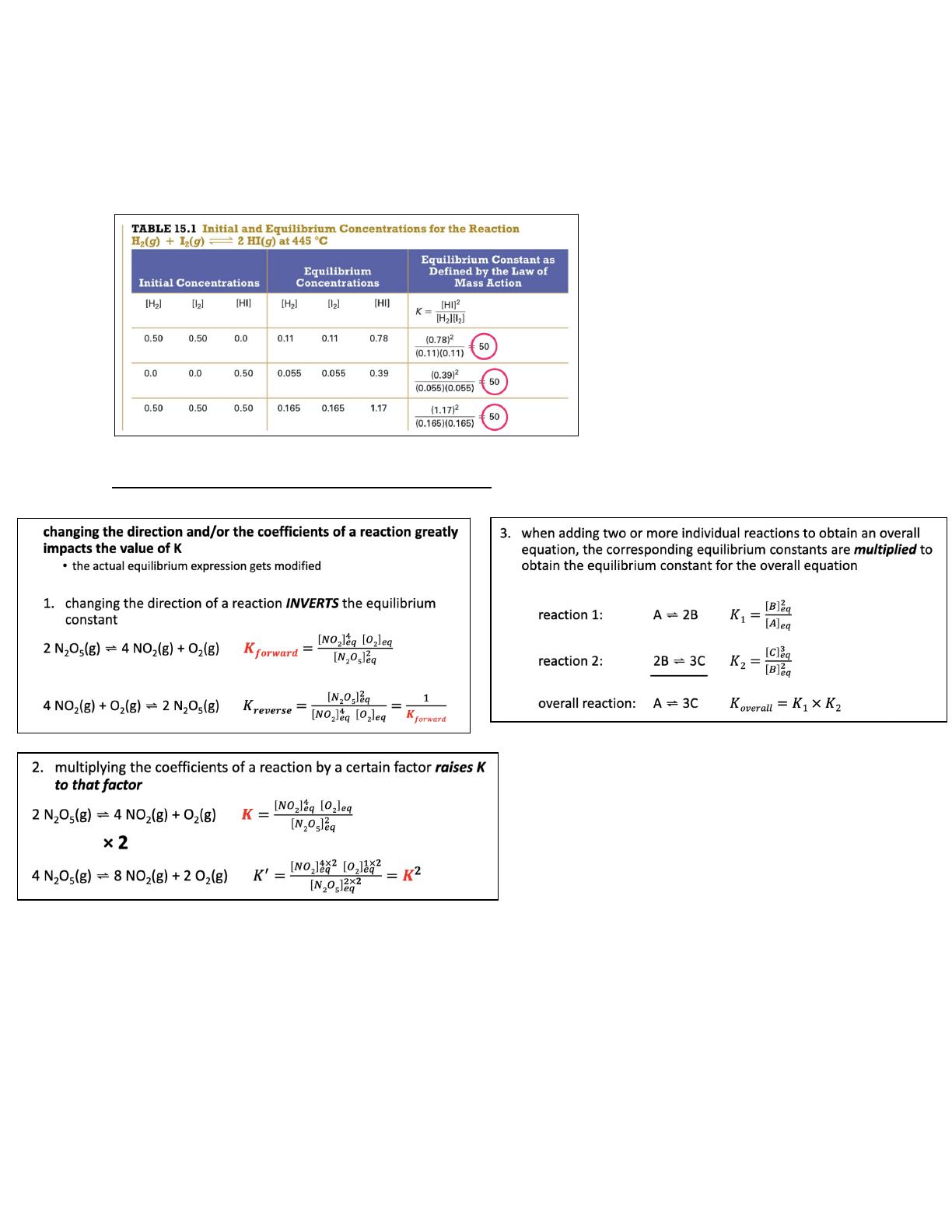
- Details the equilibrium constant (K) and its relationship to reaction concentrations.
- Covers the effects of concentration, pressure, and temperature on chemical equilibrium.
- Introduces the ICE table method for calculating equilibrium concentrations.