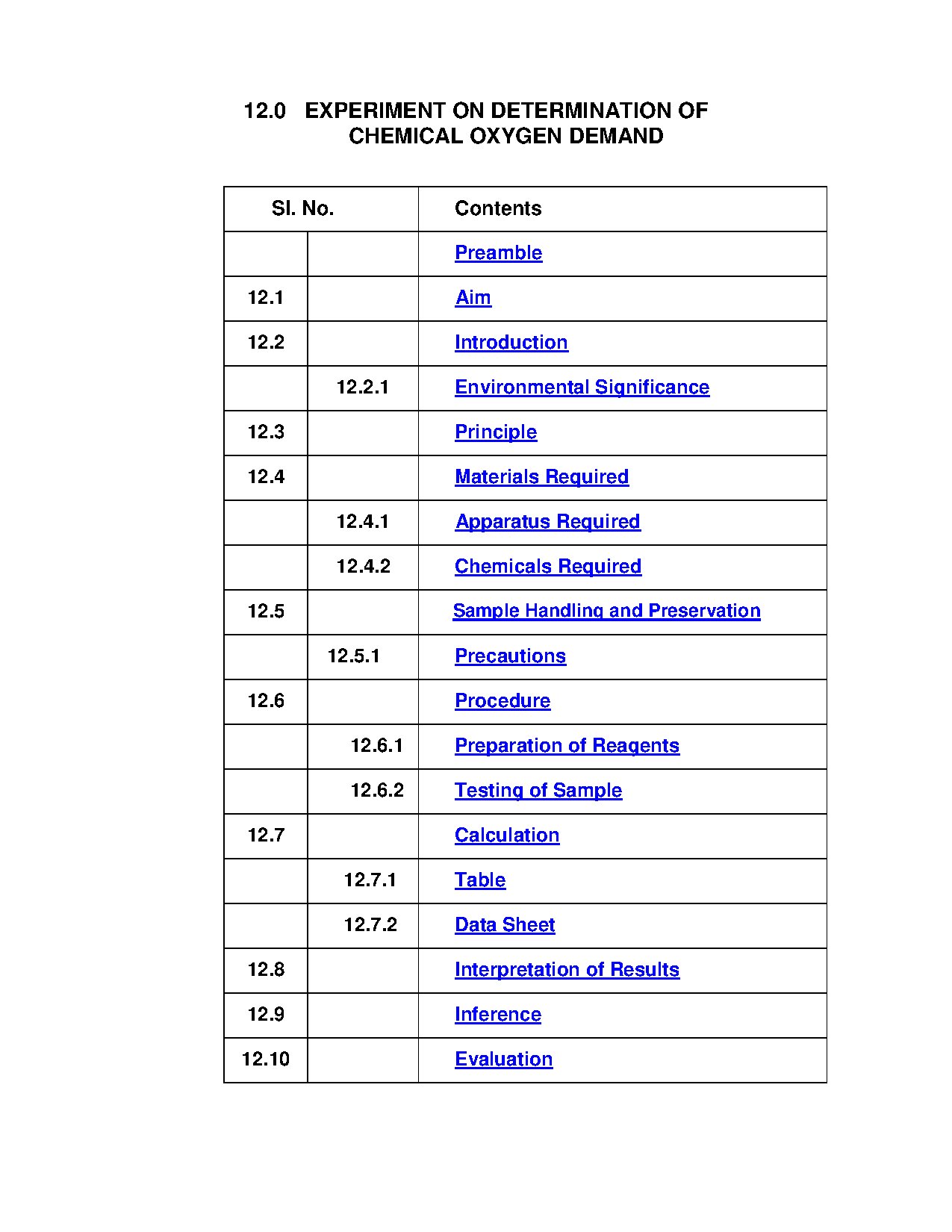
The experiment on Chemical Oxygen Demand (COD) outlines a standardized method for measuring organic pollutants in water and wastewater. It emphasizes the significance of COD in assessing water quality and its environmental implications. The procedure follows IS: 3025 (Part 58) and includes detailed steps for sample preparation, reagent usage, and titration techniques. This guide is essential for environmental science students and professionals conducting water quality assessments. It provides insights into the relationship between COD and BOD, highlighting the importance of these measurements in wastewater treatment processes.
Key Points
- Describes the procedure for determining Chemical Oxygen Demand in water samples.
- Explains the environmental significance of COD in assessing water quality.
- Details the materials and reagents required for the COD experiment.
- Includes a step-by-step guide for sample preparation and titration.
- Discusses the relationship between COD and BOD in wastewater treatment.