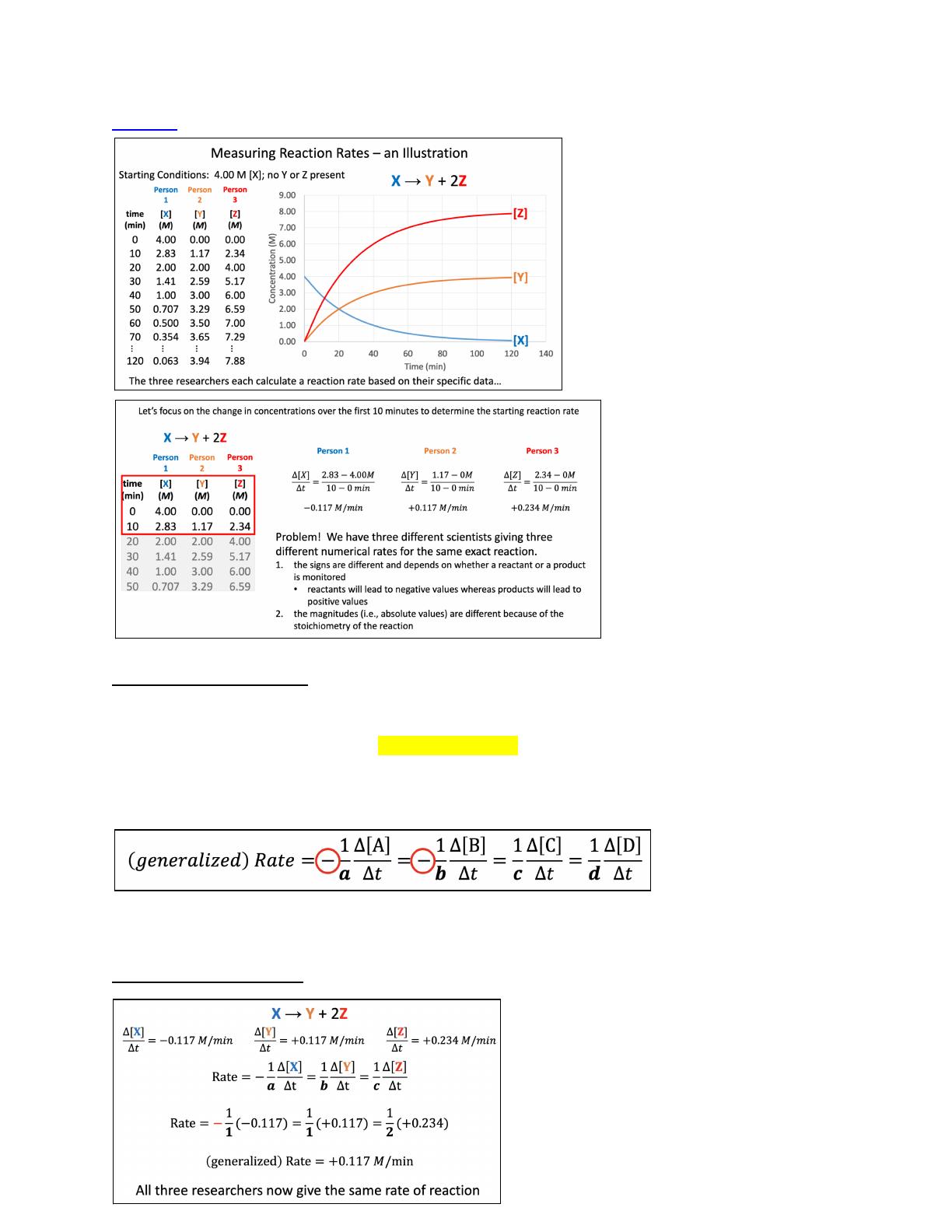
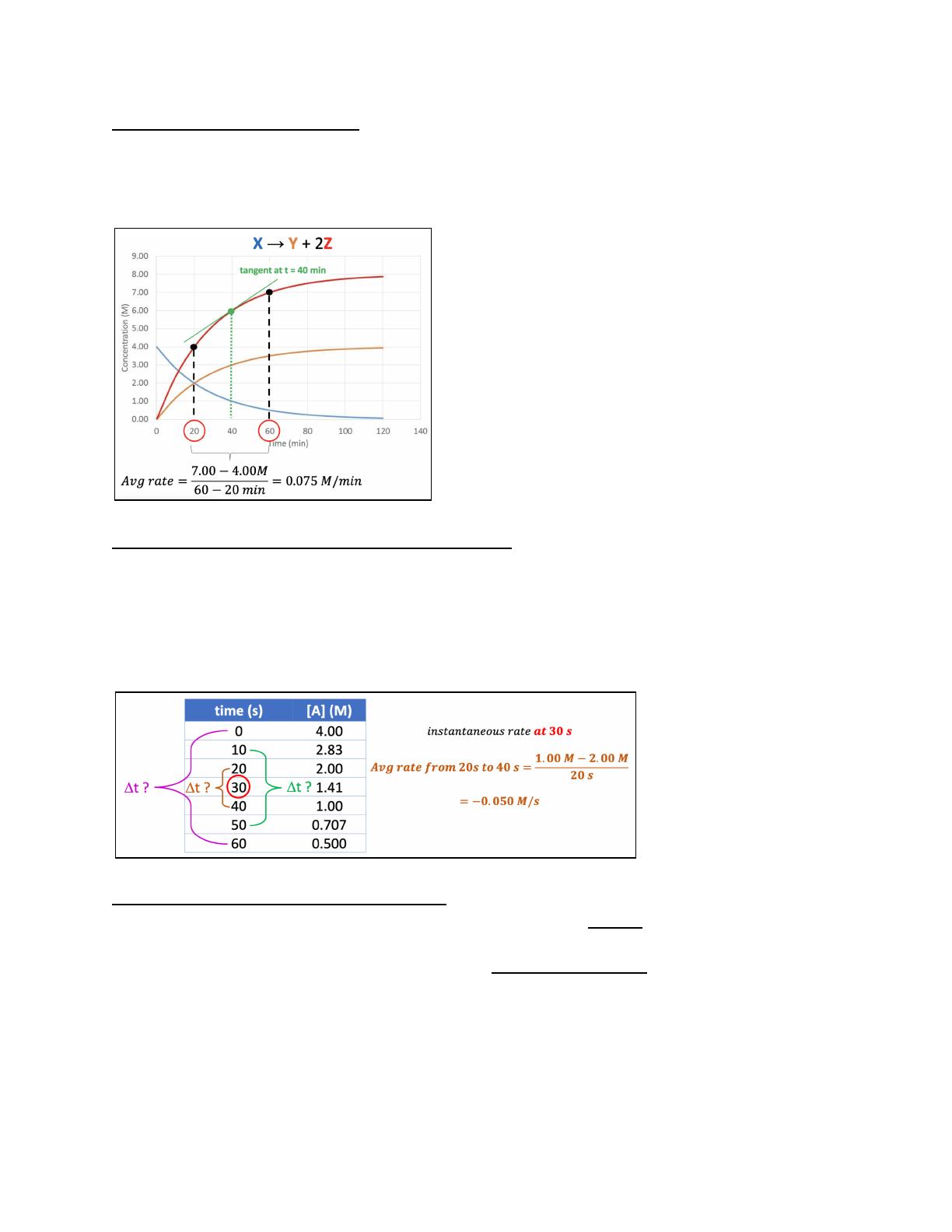
Chemical kinetics explores the rates of chemical reactions, focusing on how reactants convert to products. This chapter delves into the collision model, which posits that reactions occur when particles collide with sufficient energy and proper orientation. Key factors affecting reaction rates include reactant concentration, temperature, and molecular structure. Designed for chemistry students, this lecture provides a comprehensive overview of reaction rates, rate laws, and the impact of temperature on kinetics. It also discusses the Arrhenius equation and activation energy, essential concepts for understanding reaction mechanisms.
Key Points
- Explains the collision model of chemical kinetics and its significance in reaction rates.
- Covers factors affecting reaction rates, including concentration, temperature, and molecular orientation.
- Details the Arrhenius equation and its role in determining the rate constant for reactions.
- Discusses activation energy and its impact on the speed of chemical reactions.