Chapter 13 solutions focus on key chemistry concepts, including the definition of solutions, solubility, and the energetics of solution formation. This content is essential for chemistry students studying solution dynamics, particularly the interactions between solute and solvent. The chapter discusses factors influencing solubility, such as intermolecular forces and entropy, and provides detailed explanations of water-soluble and fat-soluble vitamins. It also covers the energetics involved in forming solutions, including endothermic and exothermic processes, making it a valuable resource for those preparing for exams in general chemistry.
Key Points
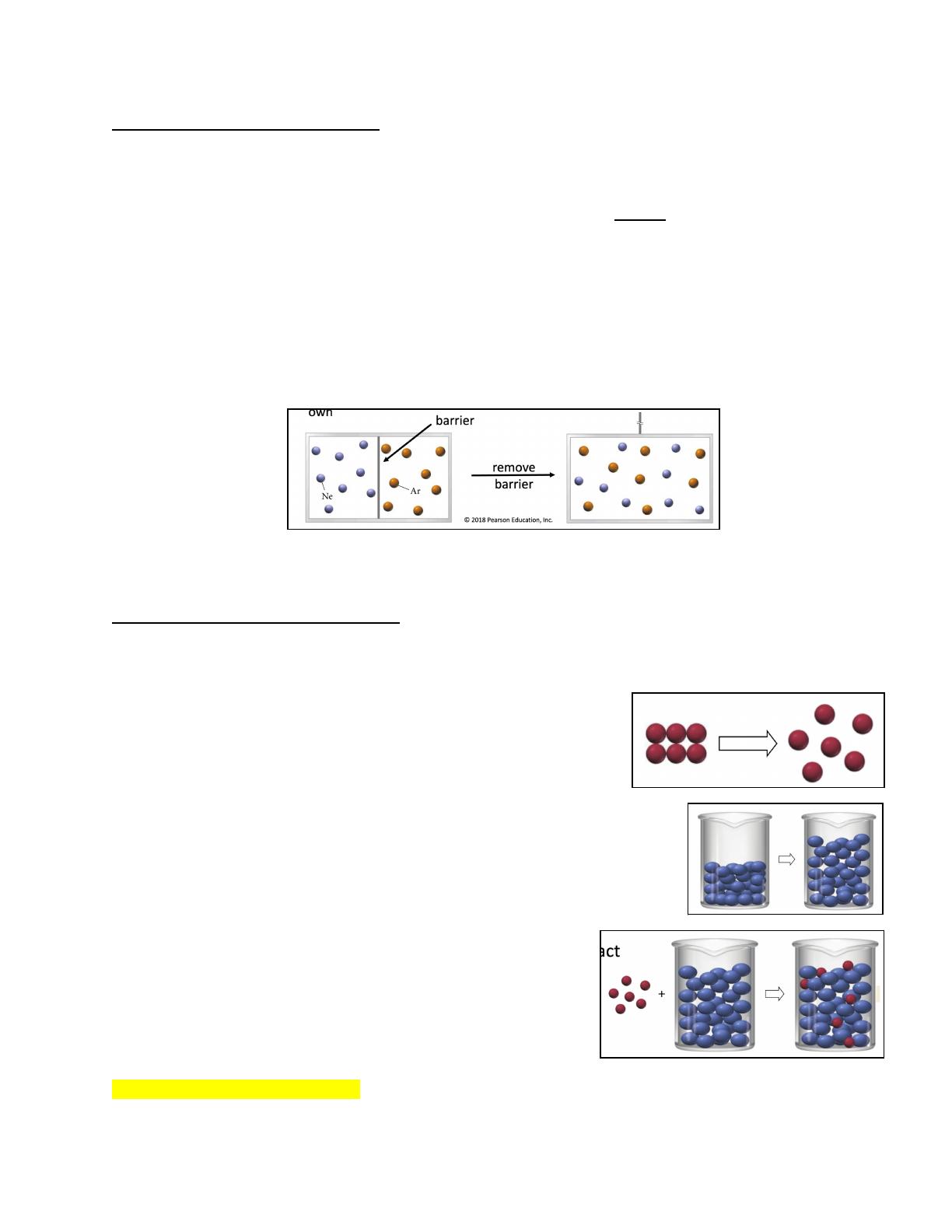
- Explains the concept of solutions as homogeneous mixtures of solute and solvent.
- Describes solubility and factors affecting it, including intermolecular forces.
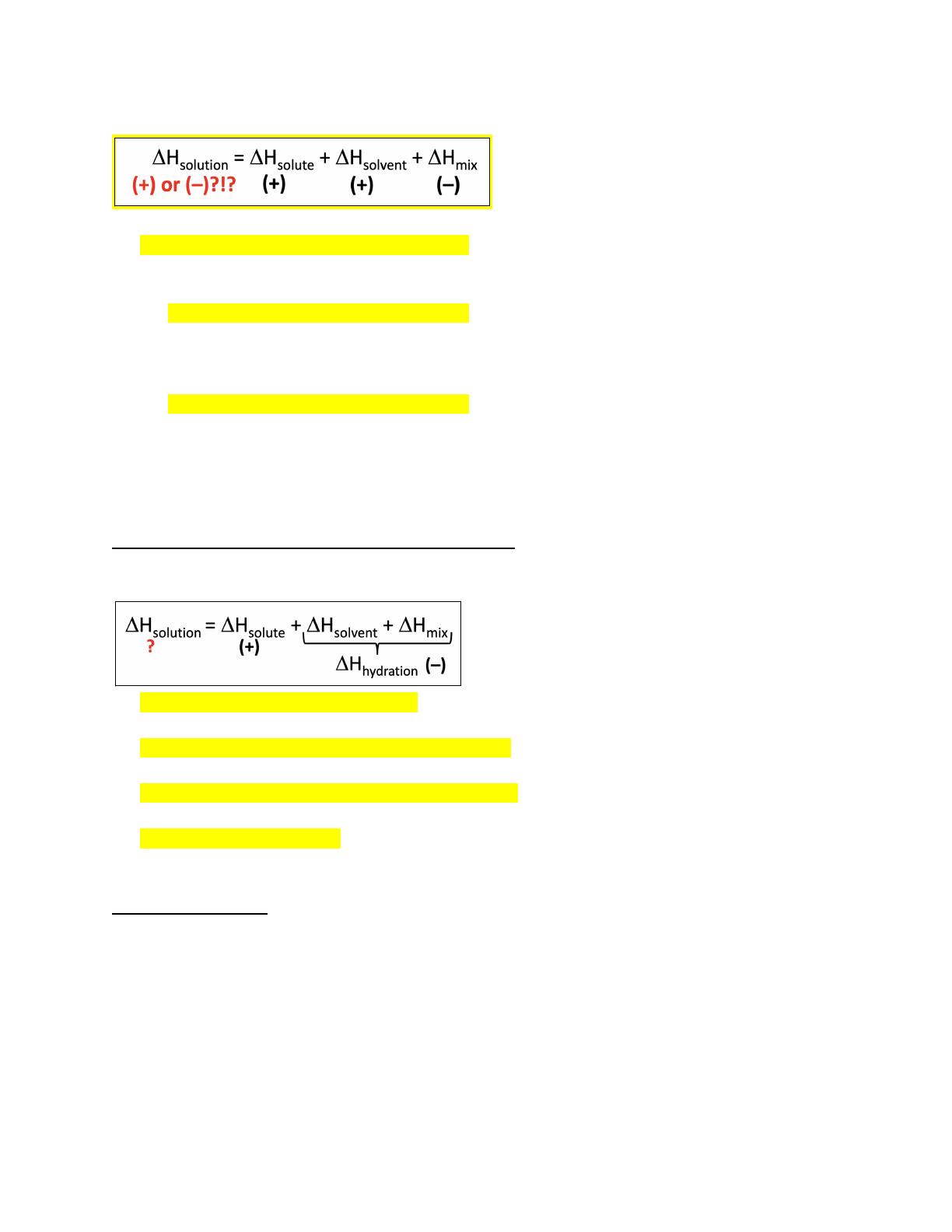
- Discusses the energetics of solution formation, including endothermic and exothermic processes.
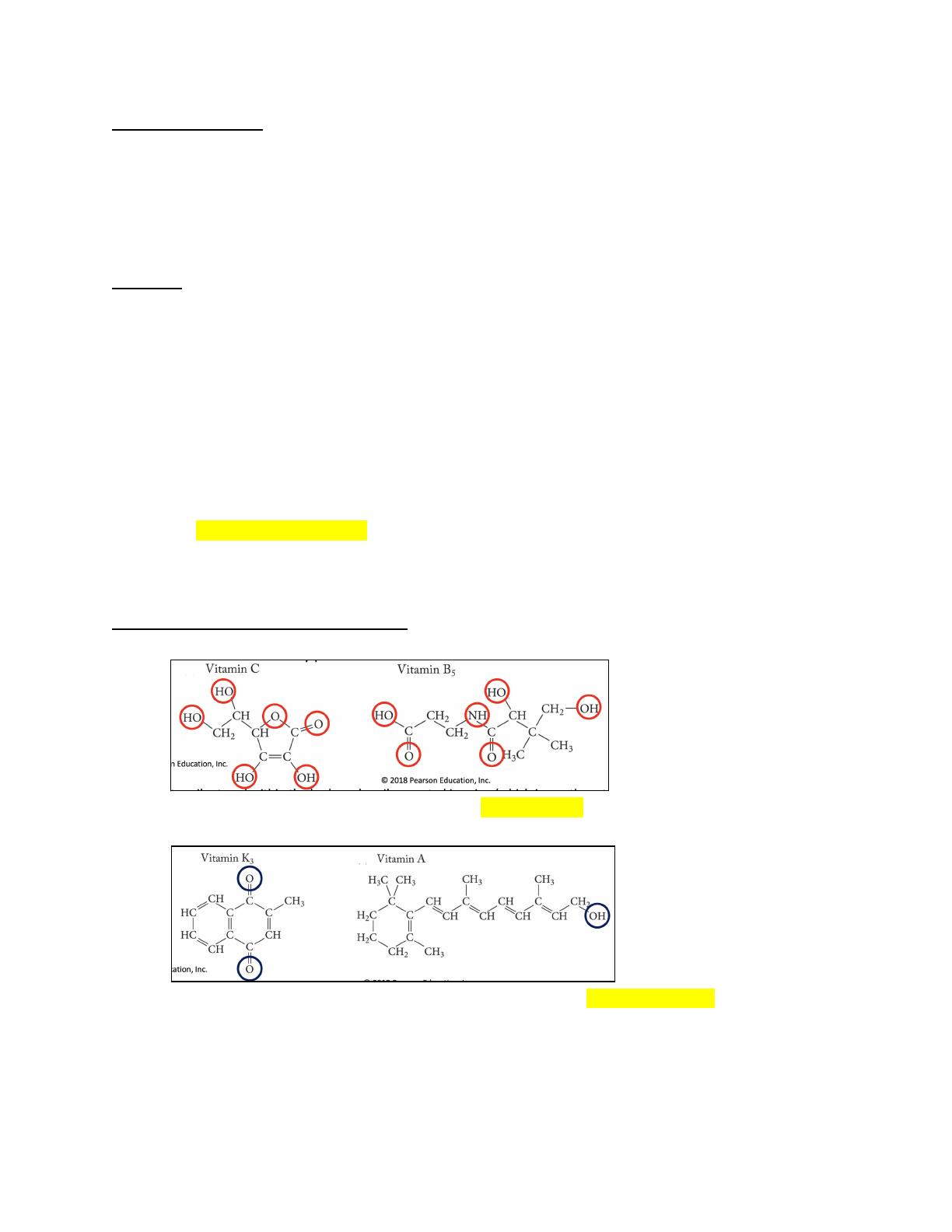
- Covers the differences between water-soluble and fat-soluble vitamins in terms of storage and excretion.