Grade 9 Chemistry Review focuses on essential concepts in chemistry, including the classification of matter, atomic structure, and the periodic table. Students will explore physical and chemical properties, changes, and the formation of ions. This comprehensive guide includes activities for drawing Bohr-Rutherford and Lewis dot diagrams, as well as practice questions on subatomic particles and the periodic table. Ideal for Grade 9 students preparing for exams or seeking to reinforce their understanding of chemistry topics.
Key Points
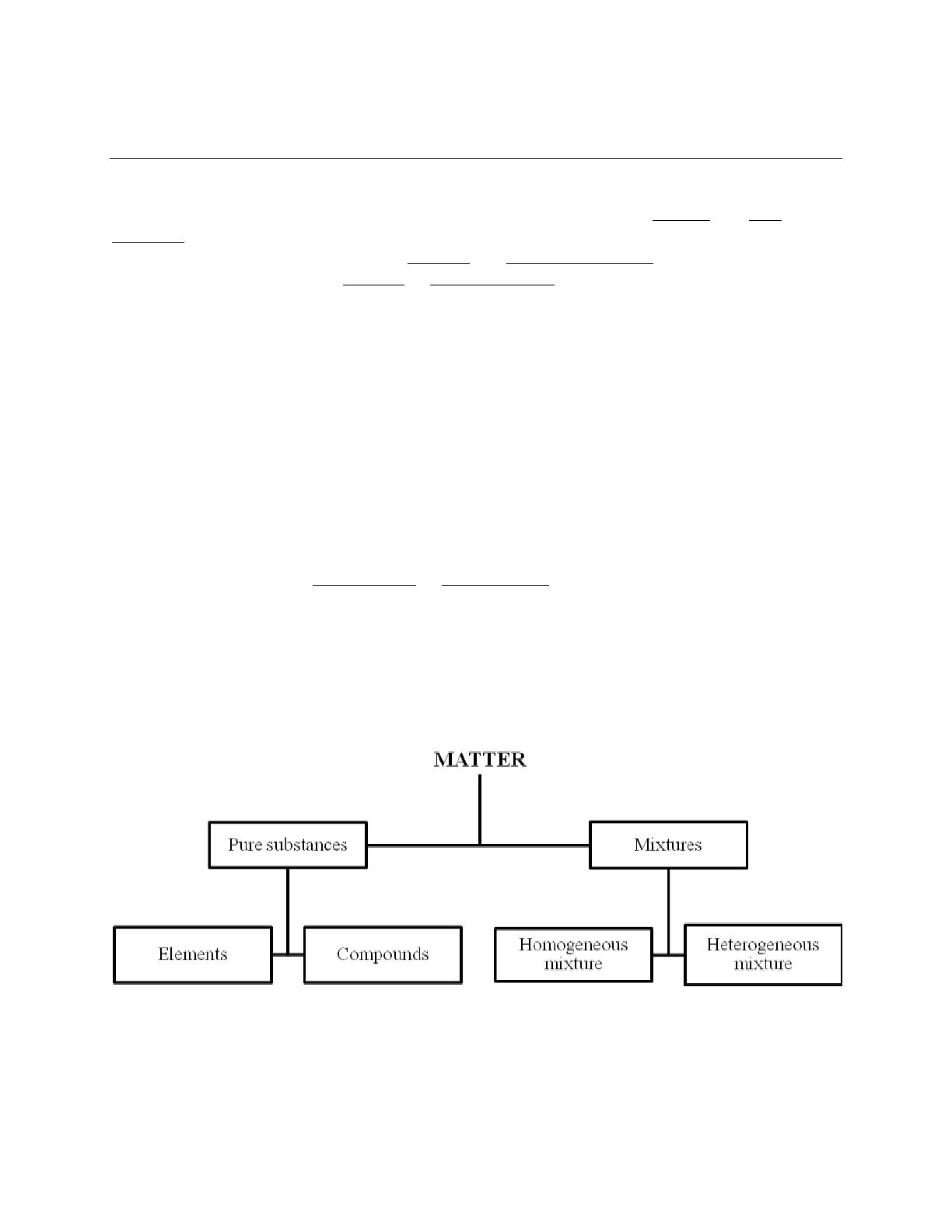
- Explains the classification of matter, including pure substances and mixtures.
- Covers atomic structure, including subatomic particles and their properties.
- Includes activities for drawing Bohr-Rutherford and Lewis dot diagrams.
- Discusses the organization of the periodic table and the properties of different element groups.