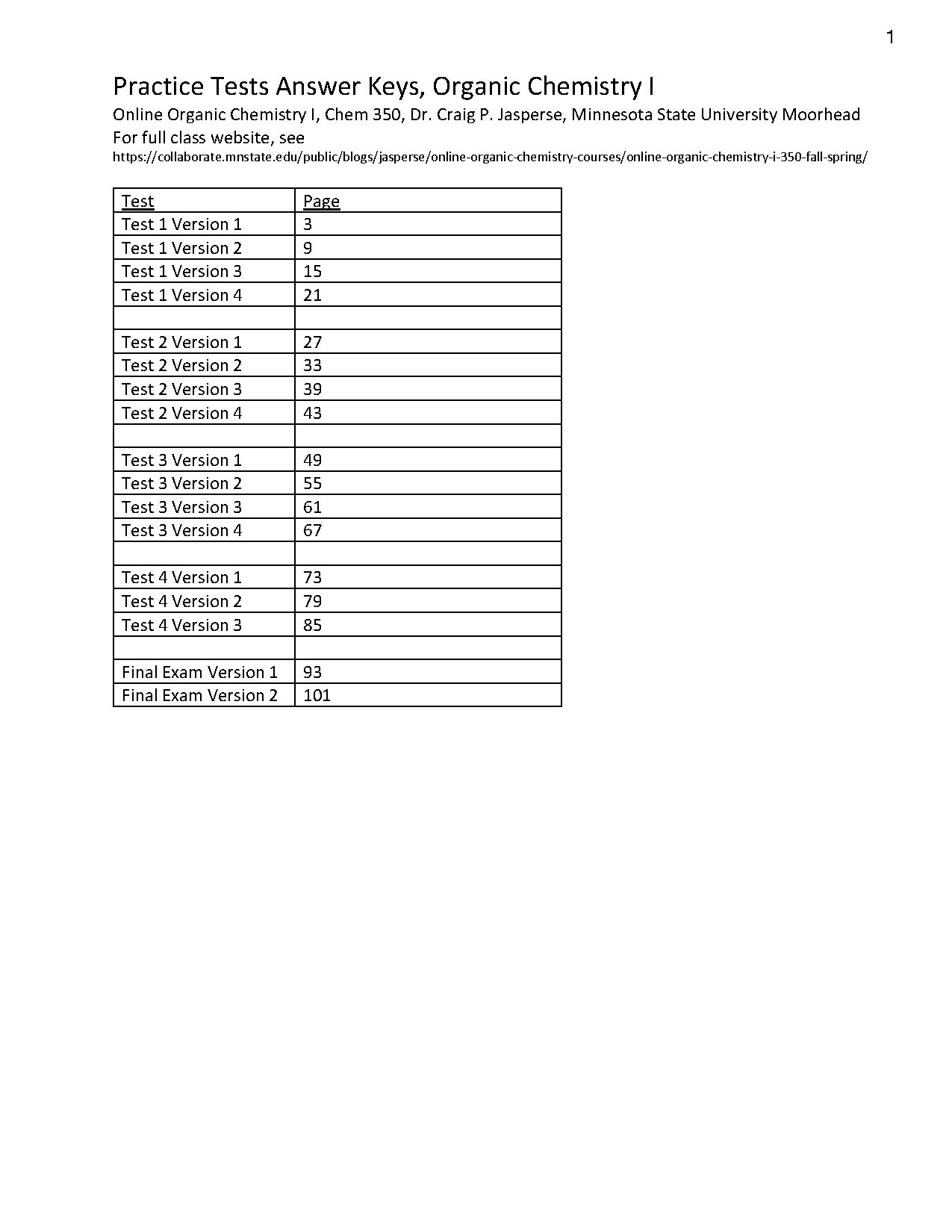
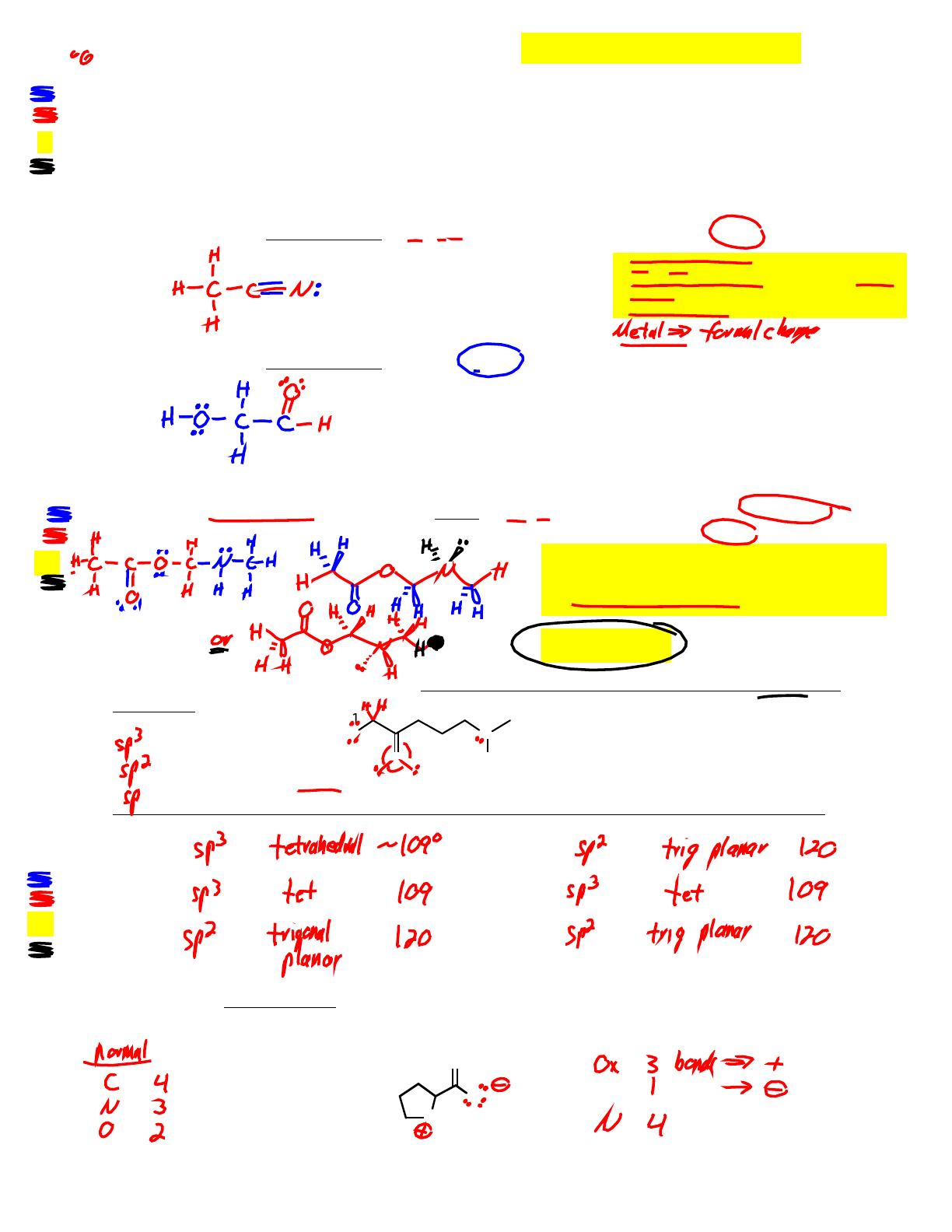
Organic Chemistry I practice tests and answer keys are essential for students enrolled in Chem 350 at Minnesota State University Moorhead. Authored by Dr. Craig P. Jasperse, this resource provides multiple versions of practice tests, including detailed answer keys for each test. Topics covered include the structure and properties of organic molecules, Lewis structures, hybridization, acidity ranking, and resonance structures. This material is ideal for students preparing for exams in organic chemistry, offering a comprehensive review of key concepts and problem-solving strategies.
Key Points
- Includes multiple versions of practice tests for Organic Chemistry I
- Covers key topics such as Lewis structures and hybridization
- Provides answer keys with detailed explanations for each test
- Ideal for Chem 350 students at Minnesota State University Moorhead