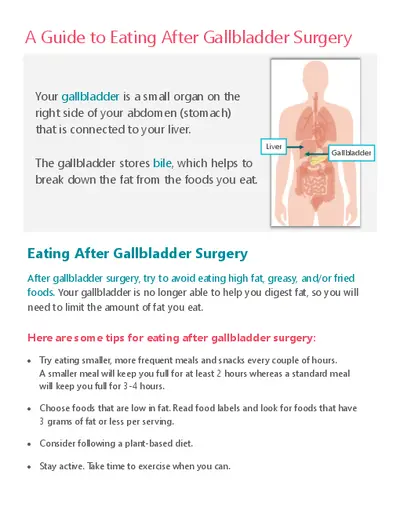
Element Builder Gizmo provides an interactive platform for students to explore atomic structure and subatomic particles. This educational resource focuses on understanding protons, neutrons, and electrons, and how they contribute to the properties of elements. Students will learn to create atoms, identify isotopes, and understand electron arrangements. Ideal for middle and high school science students, this guide enhances comprehension of fundamental chemistry concepts. Engaging activities and questions help reinforce learning and prepare students for exams in chemistry and related subjects.
Key Points
- Explains the properties and roles of protons, neutrons, and electrons in atomic structure.
- Includes interactive activities for building atoms and identifying isotopes.
- Covers electron arrangements and valence electrons critical for chemical bonding.
- Designed for middle and high school students studying chemistry concepts.