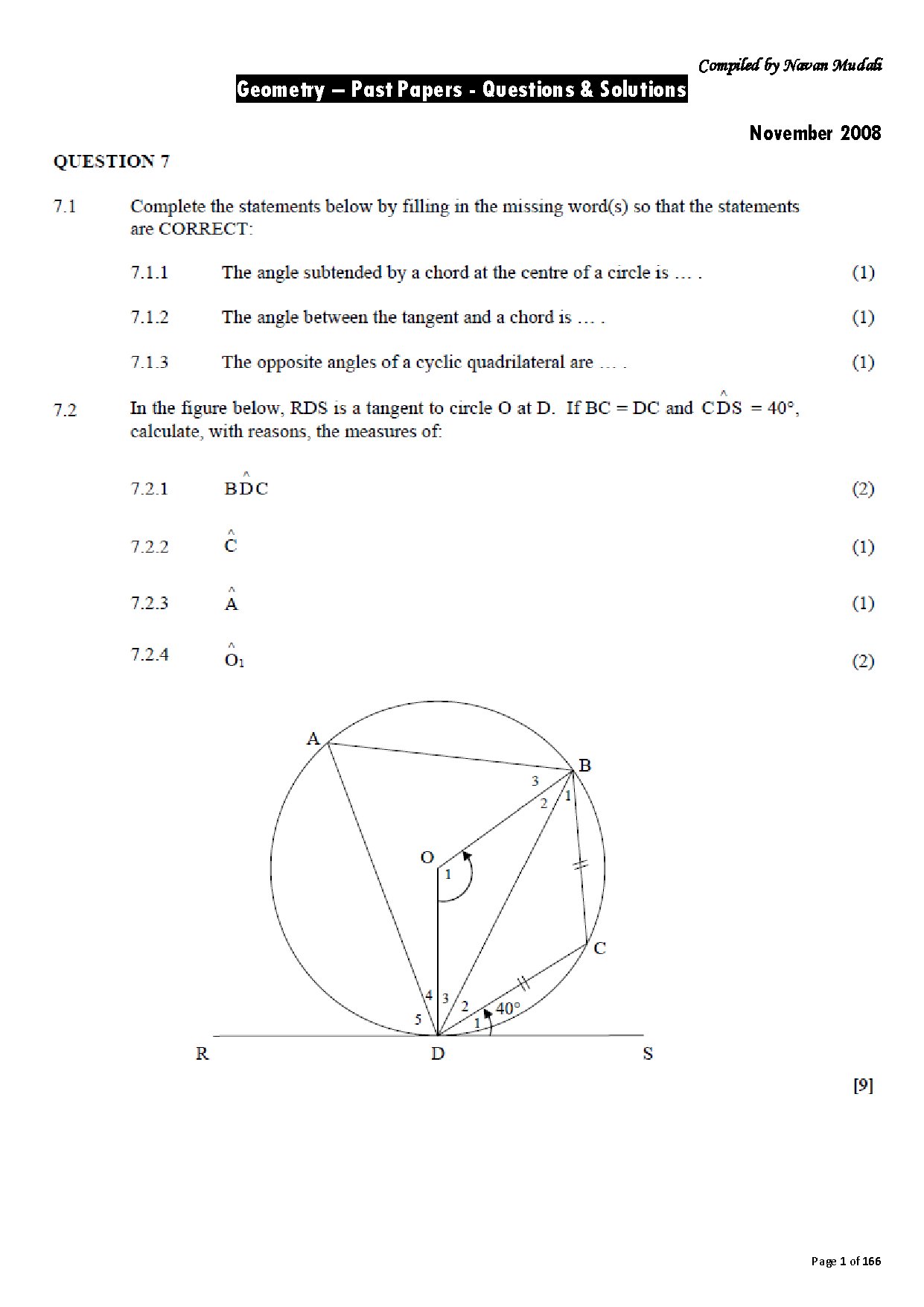
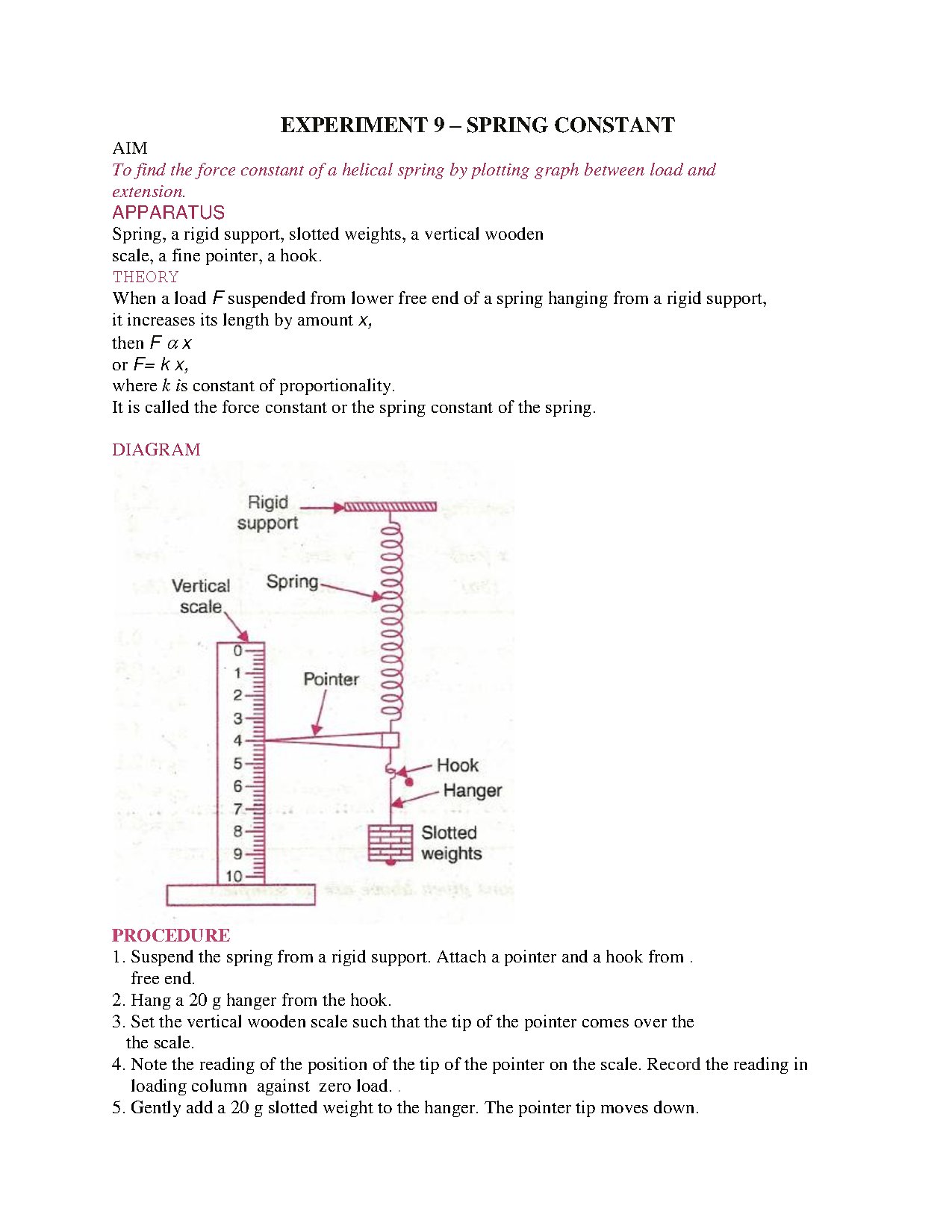
Labs 2 & 3 (Chem 2070) focus on foundational quantitative techniques: measuring mass, length, and volume using SI units; mastering metric conversions; calculating density; and applying significant figures. Students perform unit conversions (e.g., in. to cm, mL to L) and analyze data from instruments like graduated cylinders, essential for future chemical analysis.