within a common chemical framework. For the sake of
clarity, in this book we sometimes risk certain general-
izations, which, though not perfect, remain useful; we
also frequently point out the exceptions that illuminate
scientific generalizations.
Biochemistry describes in molecular terms the struc-
tures, mechanisms, and chemical processes shared by
all organisms and provides organizing principles that
underlie life in all its diverse forms, principles we refer
to collectively as the molecular logic of life. Although
biochemistry provides important insights and practical
applications in medicine, agriculture, nutrition, and
industry, its ultimate concern is with the wonder of life
itself.
In this introductory chapter, then, we describe
(briefly!) the cellular, chemical, physical (thermody-
namic), and genetic backgrounds to biochemistry and
the overarching principle of evolution—the develop-
ment over generations of the properties of living cells.
As you read through the book, you may find it helpful
to refer back to this chapter at intervals to refresh your
memory of this background material.
1.1 Cellular Foundations
The unity and diversity of organisms become apparent
even at the cellular level. The smallest organisms consist
of single cells and are microscopic. Larger, multicellular
organisms contain many different types of cells, which
vary in size, shape, and specialized function. Despite
these obvious differences, all cells of the simplest and
most complex organisms share certain fundamental
properties, which can be seen at the biochemical level.
Cells Are the Structural and Functional Units of All
Living Organisms
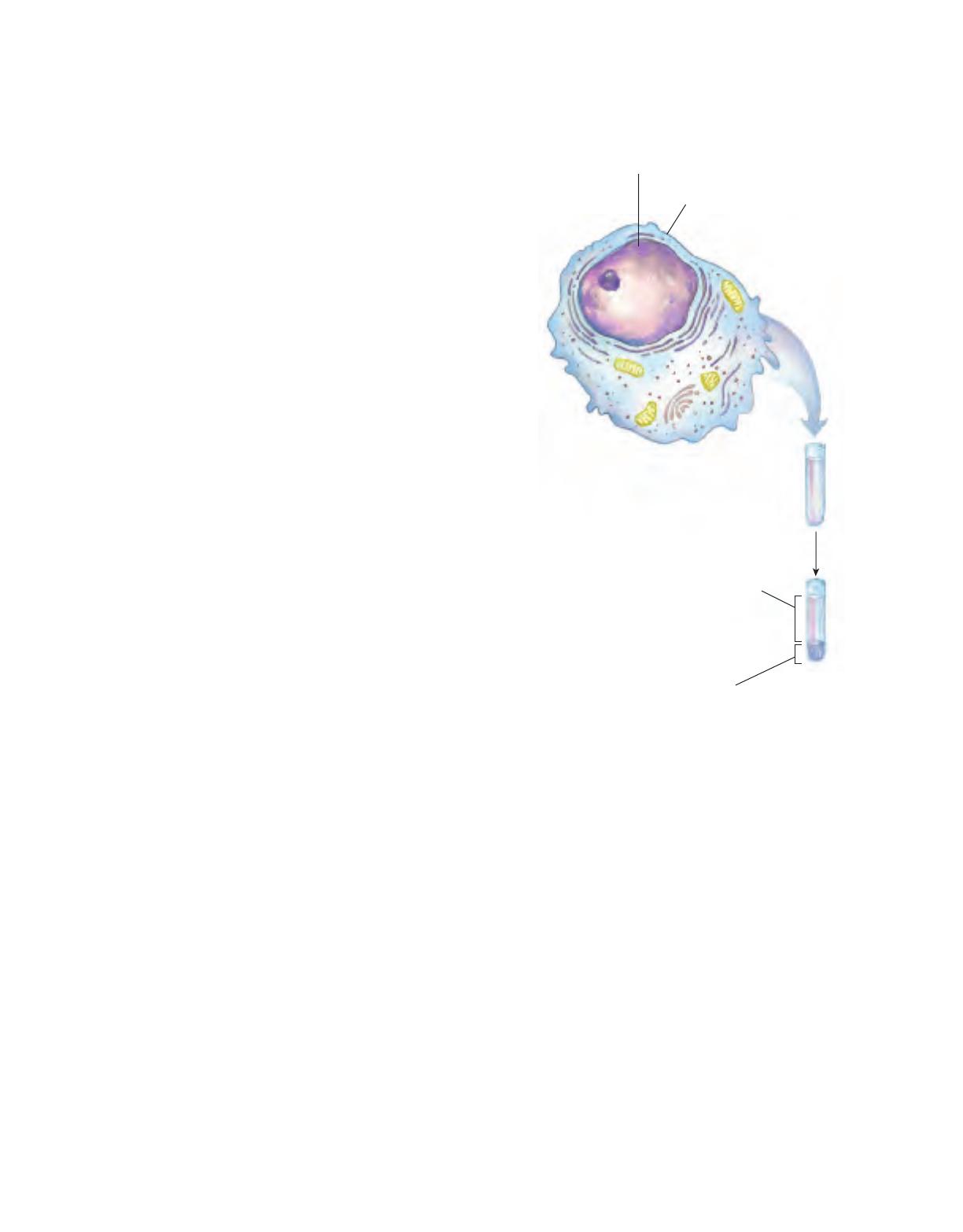
Cells of all kinds share certain structural features (Fig.
1–3). The plasma membrane defines the periphery of
the cell, separating its contents from the surroundings.
It is composed of lipid and protein molecules that form
a thin, tough, pliable, hydrophobic barrier around the
cell. The membrane is a barrier to the free passage of
inorganic ions and most other charged or polar com-
pounds. Transport proteins in the plasma membrane al-
low the passage of certain ions and molecules; receptor
proteins transmit signals into the cell; and membrane
enzymes participate in some reaction pathways. Be-
cause the individual lipids and proteins of the plasma
membrane are not covalently linked, the entire struc-
ture is remarkably flexible, allowing changes in the
shape and size of the cell. As a cell grows, newly made
lipid and protein molecules are inserted into its plasma
membrane; cell division produces two cells, each with its
own membrane. This growth and cell division (fission)
occurs without loss of membrane integrity.
The internal volume bounded by the plasma mem-
brane, the cytoplasm (Fig. 1–3), is composed of an
aqueous solution, the cytosol, and a variety of sus-
pended particles with specific functions. The cytosol is
a highly concentrated solution containing enzymes and
the RNA molecules that encode them; the components
(amino acids and nucleotides) from which these macro-
molecules are assembled; hundreds of small organic
molecules called metabolites, intermediates in biosyn-
thetic and degradative pathways; coenzymes, com-
pounds essential to many enzyme-catalyzed reactions;
inorganic ions; and ribosomes, small particles (com-
posed of protein and RNA molecules) that are the sites
of protein synthesis.
All cells have, for at least some part of their life, ei-
ther a nucleus or a nucleoid, in which the genome—
1.1 Cellular Foundations
3
Nucleus (eukaryotes)
or nucleoid (bacteria)
Contains genetic material–DNA and
associated proteins. Nucleus is
membrane-bounded.
Plasma membrane
Tough, flexible lipid bilayer.
Selectively permeable to
polar substances. Includes
membrane proteins that
function in transport,
in signal reception,
and as enzymes.
Cytoplasm
Aqueous cell contents and
suspended particles
and organelles.
Supernatant: cytosol
Concentrated solution
of enzymes, RNA,
monomeric subunits,
metabolites,
inorganic ions.
Pellet: particles and organelles
Ribosomes, storage granules,
mitochondria, chloroplasts, lysosomes,
endoplasmic reticulum.
centrifuge at 150,000 g
FIGURE 1–3 The universal features of living cells. All cells have a
nucleus or nucleoid, a plasma membrane, and cytoplasm. The cytosol
is defined as that portion of the cytoplasm that remains in the super-
natant after centrifugation of a cell extract at 150,000 g for 1 hour.
8885d_c01_003 12/20/03 7:03 AM Page 3 mac76 mac76:385_reb:




