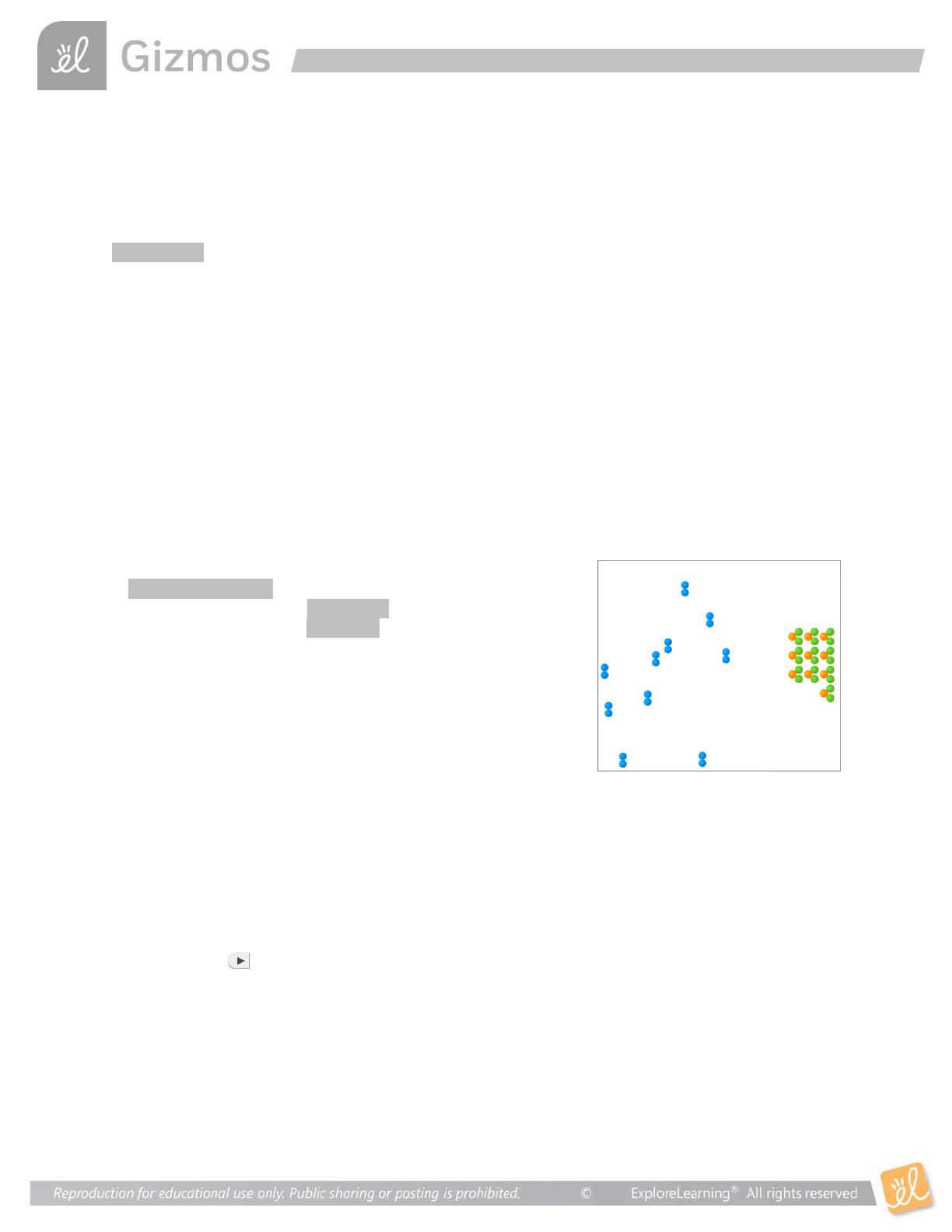
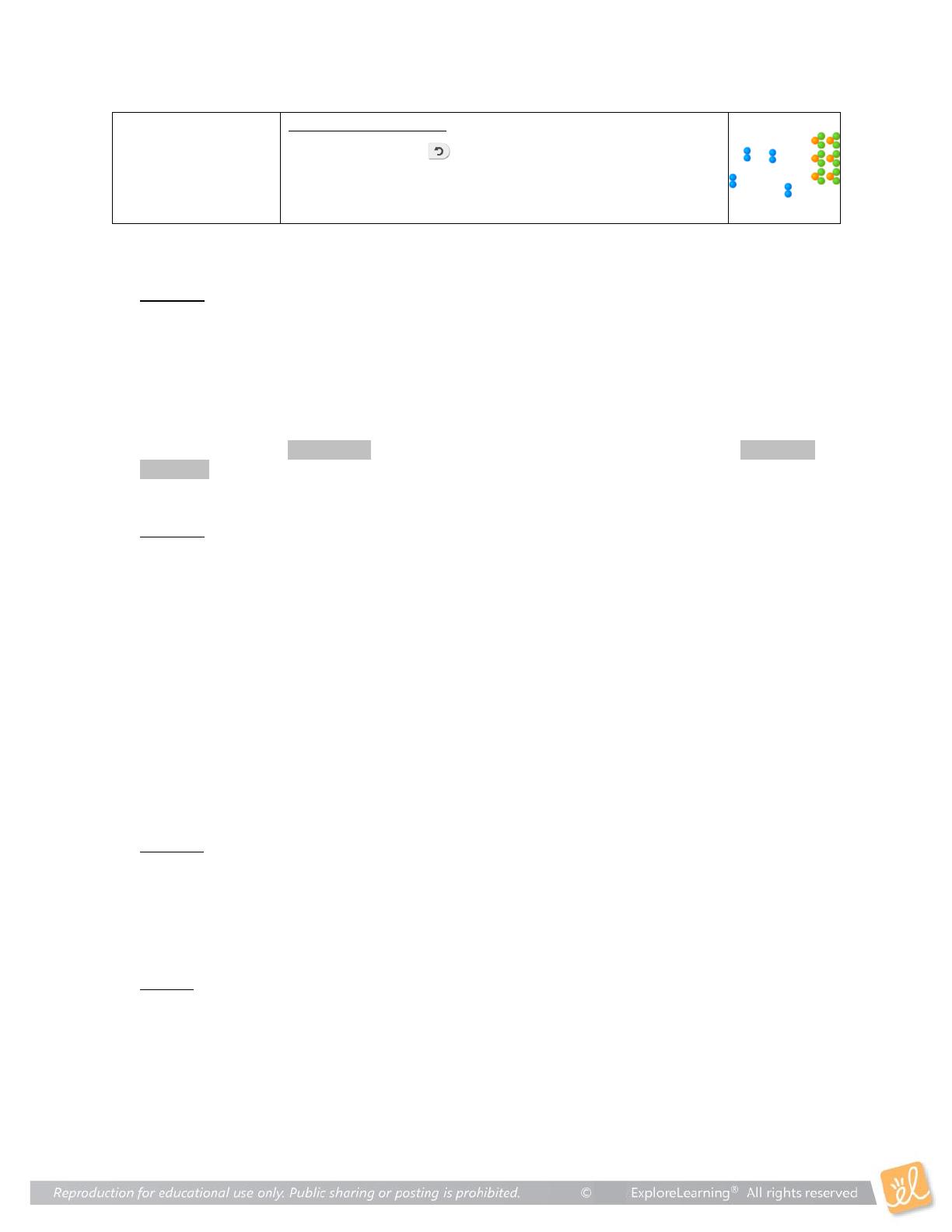
Collision Theory explores the principles of chemical reactions, focusing on how temperature, surface area, and concentration affect reaction rates. Designed for high school chemistry students, this resource includes hands-on activities and simulations to visualize molecular interactions. Key concepts include the activated complex, catalysts, and the importance of molecular collisions. Ideal for educators and students preparing for exams, this exploration provides a comprehensive understanding of reaction dynamics.
Key Points
- Explains the Collision Theory and its significance in chemical reactions.
- Includes hands-on activities to visualize the effects of temperature on reaction rates.
- Covers the role of catalysts in accelerating chemical reactions.
- Analyzes how surface area and concentration influence reaction dynamics.